Research focus Lang labor
In particular, we analyse how host molecules influence virus replication in different cell populations. Strong suppression of viral replication (e.g.: in Kupffer cells, red pulp macrophages) is important to prevent the spread of viruses and chronic virus infection. In contrast, there are cells in secondary lymphatic organs (e.g.: CD169+ macrophages and dendritic cells) that deliberately allow virus replication. This "forced viral replication" is important for efficient immune activation in some viruses.
Which molecules influence virus replication, in which cells these molecules are active and how they cause immune activation and/or immunopathology is a laboratory project.
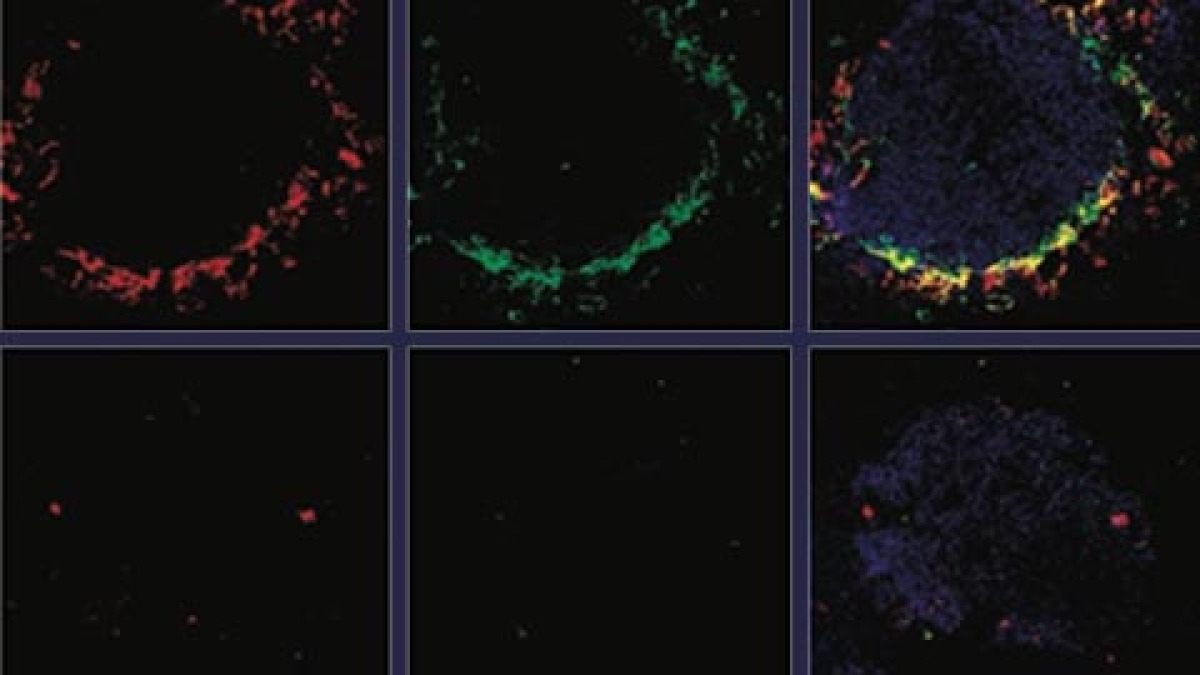
In recent years we study the how the cell-type specific replication of virus influences immunity, pathogenicity and/or persistence of virus infection. We found that antigen-presenting cells express proteins (i.e. UBP43, Usp18) which allows them to replicate a broad range of viruses. By doing so they generate huge amounts of viral antigens and pattern recognition ligands, which than induce a fast immune action. In contrast afferent macrophages within the lymph node as well as red pulp macrophages (spleen) and Kupffer cells (liver) have strongly antiviral capacity. While such macrophage can take up huge amounts of virus, the virus cannot infect the macrophage and is degraded. In a recent publication we identified acid ceramidase and sphingosine as one key parameter in this process.
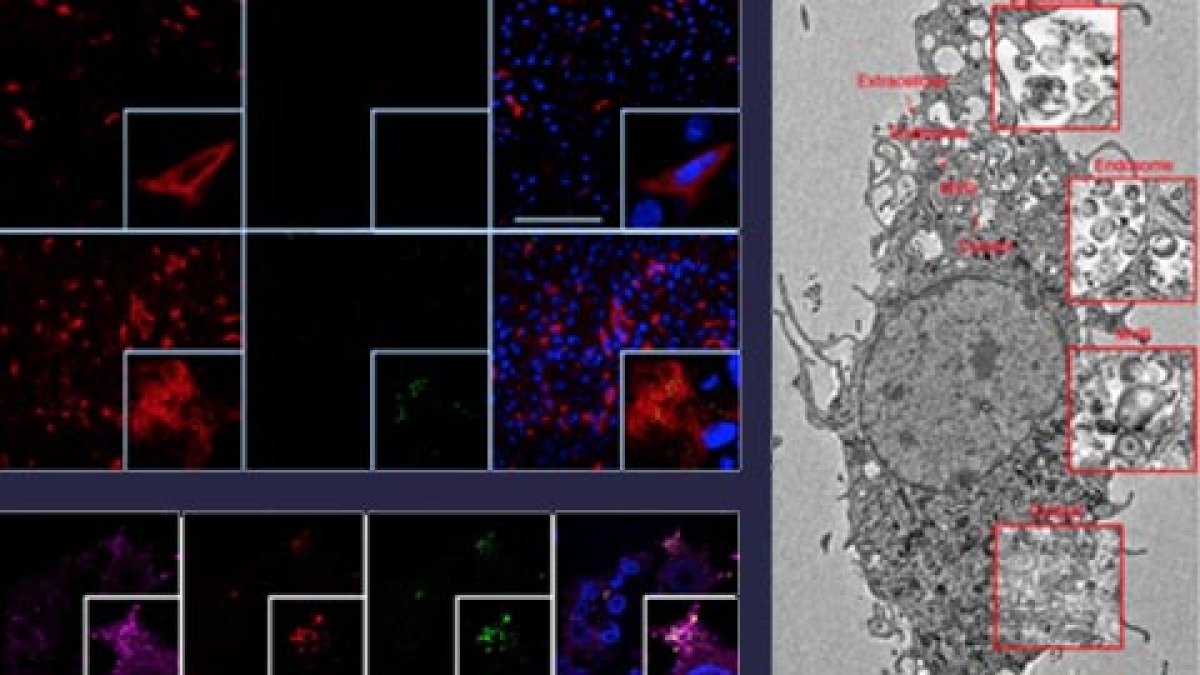
Within a model of infection with the Herpes simplex virus (HSV) we found that macrophages take up HSV-1 via endocytosis and transport the virions into multi-vesicular bodies (MVBs). In MVBs, acid ceramidase (aCDase) converts ceramide into sphingosine and increases the formation of sphingosine-rich intraluminal vesicles (ILVs). Once HSV-1 particles reach MVBs, sphingosine-rich ILVs bind to HSV-1 particles, which restricts fusion with the limiting endosomal membrane and prevents cellular infection. Lack of aCDase in macrophage cultures or in Asah1–/– mice results in replication of HSV-1 and Asah1–/– mice die soon after systemic or intravaginal inoculation. The treatment of macrophages with sphingosine enhancing compounds blocks HSV-1 propagation, suggesting a therapeutic potential of this pathway. In direct comparison Asah1 had similar antiviral activity as type I interferon and the innate signalling molecules MyD88, Trif and Cardif.


