Electrochemistry of metals
Electrochemistry of metals
Depending on the needs of our actual research you will perform in this experiment cyclovoltammetry of metals. The aim of this experiment is to familiarize you with the basics of metals's electrochemistry using a catalytically active standard system. In one our experiment, this is the platinum-sulphuric acid system. We use this system to study the adsorption of atomic hydrogen. A brief overview of the chemical reactions can be found in section
However, sometimes we change this experiment's topic to for example oxidation of Nickel, Titanium or Rhodium.
The difference to the experiment electrochemistry 2 is that the adsorbed species (in most cases hydrogen or oxygen) is directly adsorbed on the metal electrode.
Later we will collect the electrochemical data of our master students of many metals. But at first we will introduce you to platinum's electrochemistry.
Platinum in electrochemistry
Please read a little bit about the special role of platinum in electrochemistry. This article in german introduces you the fundamentals of platinums electrochemistry. You can also read it with some more details in english.
At the end of the 1980's platinums electrochemistry became very popular again due to the availibality of platinum single crystal electrodes. Please have a look to the cyclovoltammograms in this paper. The peaks are pretty narrow and well defined when you compare it with the cyclovoltammograms in Bölts paper.
In our paper the mentioned flame annealing in the paper of the Markovic group will play a vital role. We will also use it produce small electrode for the production of small defined electrode as in the paper of Tavares.
Hydrogen adsorption on platinum
Hydrogen can adsorb in acidic solution from the oxonium ion.
$$ \mathrm{H_{3}O^{+} + M + e^{-}_{metall} \rightarrow MH_{ad} + H_{2}O }$$
With this notation M means a free adsorption site on the metal surface
In neutrale or alkaline solution the adsorption from the $ \mathrm{H_{2} O } $ molecule is prevalent.
$$ \mathrm{ H_{2}O + M + e^{-}_{metall} \rightarrow MH_{ad} + OH^{-} } $$.
As a second reaction step the recombination reaction of two adsorbed species may occur:
$$ \mathrm{ 2 MH_{ad} \rightarrow H_{2} + 2 M } $$
This reaction is named after Julius Tafel, a Switzerland born electrochemist furtheron active in germany
Julius Tafel published the Tafel equation of electrochemical kinetics named after him in 1905.
A reaction with water species close to the electrode is also possible.
$$ \mathrm{ MH_{ads} + H_{3}O^{+} + e^{-}_{metall} \rightarrow H_{2} + H_{2}O + M } $$
This reaction is named after Jaroslav Heyrosvksy developed the electrochemical analysis method of polarography using the mercury drop electrode in 1925 and was awarded the Nobel Prize for Chemistry in 1959. From 1950 he was head of the newly founded Polarography Institute at Charles University in Prague.
The electrochemistry of gold in alkaline solution
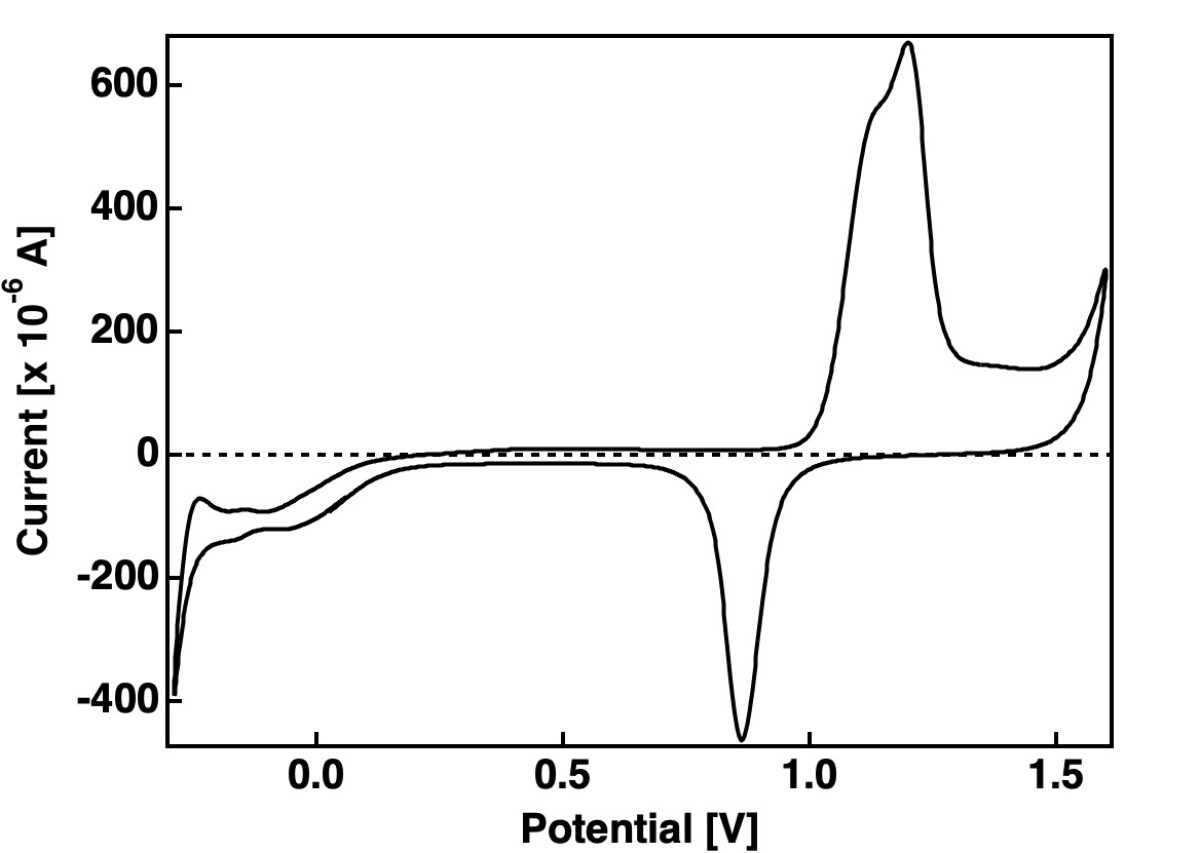
Raw data of gold
In the graph you find the cyclovoltammogram of a polycrystalline gold electrode. Besides the CV are taken with a scan rate of 50 mV/s in 0.1 N sulphuric acid electrolyte. The chemical details and the parameters of data monitoring are in the header of the the csv file. You see that our data look pretty ugly, the data are not smoothed by a software procedure. This ugly appearance is pretty normal for an electrochemical cell without a Faraday shielding. Please always show in your lab report at first the untreated data.
Treated data of gold
And here you find textbook like data which are also achievable in our lab. So nice, but do not forget to mention the smoothing factor. In this case it was chosen as 111. Further discussion in the lab. Data on GitHub.
If your interested in a microscopic interpretration of gold's data in acide solution you can have a look here.