Research
Research
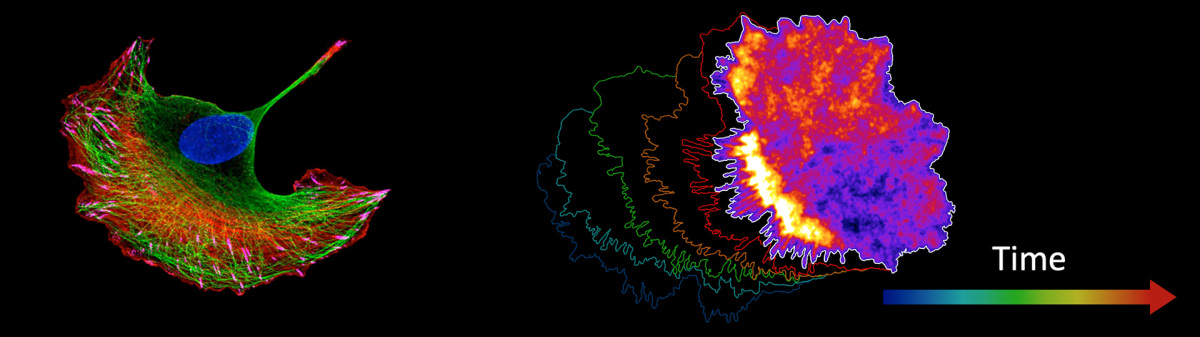
Emergence of Spontaneous Pulses and Propagating Contraction Waves
While the control of the cytoskeleton by Rho GTPase signaling is often depicted with a simple, linear pathway, recent studies uncovered non-linear, feedback-driven mechanisms that can control cytoskeletal dynamics in space and time. In our studies of adherent interphase cells, we uncovered an excitable RhoA signaling network that generates spontaneous pulses and traveling waves of cell contraction.
This network couples fast positive feedback between active RhoA and Lbc-type GEFs with delayed negative feedback mediated by Myosin II activity (Graessl et al, 2017; Kamps et al, 2020). These dynamics produce dynamic, subcellular contraction pulses and wave-like activity patterns within the cell cortex. Interestingly, we observed that matrix stiffness modulates the frequency of these contractions, suggesting that this signaling architecture may act as a mechanosensitive module, linking extracellular matrix cues to intracellular contractile responses.
Movie: Graessl et al.
Control of Directional Cell Migration
Directional cell migration depends on the precise coordination between actin-based protrusions at the leading edge and actomyosin-based contractile forces at the rear. The mechanism, how these processes are coordinated in space and time, is still poorly understood.
In recent work, we investigated the interplay between Rac1 and RhoA, two key regulators of cell migration. Rac1 promotes membrane protrusions, while RhoA drives actomyosin contraction. Although traditionally viewed as antagonists, our studies in the epidermoid carcinoma cell line A431 revealed that Rac1 can locally stimulate RhoA activity. This feedback is essential for coordinating dynamic protrusion–retraction cycles and supports an exploratory mode of migration, characterized by frequent changes in direction (Nanda et al, 2023).
In contrast, untransformed cells often display more persistent migration. Our work in Normal Human Epidermal Keratinocytes showed that the Rho GTPase Cdc42 is strongly activated at the cell rear, where it promotes sustained myosin-mediated contraction. This rearward localization of Cdc42 activity was surprising, as this molecule is classically viewed as a cell protrusion regulator at the front of migrating cells (Patwardhan et al., 2023). In the primary human keratinocytes, Cdc42 appears to stabilize polarity and reinforce directional persistence from the back of the cell, suggesting a distinct regulatory logic from that observed in carcinoma cells.
Movie: Partwardhan et al.


