FOR 1993 - Subproject GM2
Exergetic analysis of reactive systems for polygeneration
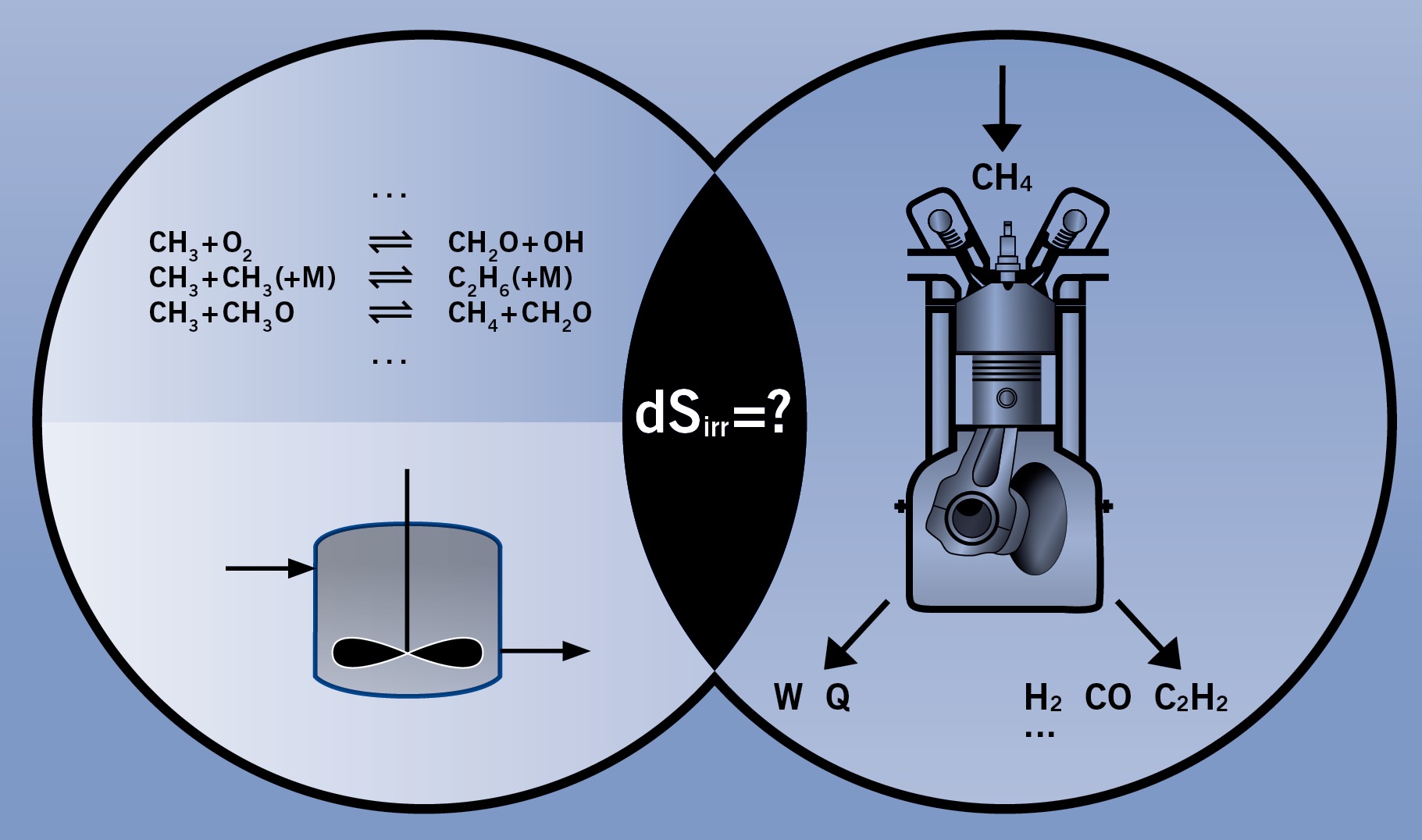
In this project reaction paths within internal combustion engines which will be designed to produce work, heat and base chemicals will be analyzed thermodynamically. Such processes are known as polygeneration processes Determination of the exergy (availability) losses of different alternative concepts and the related process parameters provides a consistent method of assessing the value of the concepts. In addition, the balances of energy and availability for the motor projects (MM1 and MM2) within the research unit will be calculated using the experimental data. This will help to validate the models. Methane will be investigated as the first fuel, and synthesis gas (CO/H2) and higher alkenes will be analyzed as possible useful products. The calculations will initially follow equilibrium paths and subsequently be continued along kinetic paths as identified by predictive elementary reaction mechanisms. The main goal is to provide second law efficiencies for high temperature polygeneration concepts. Additionally, the thermodynamic analysis is expanded by exergoeconomic analyses, to gain insights in the profitability of the polygeneration concept.
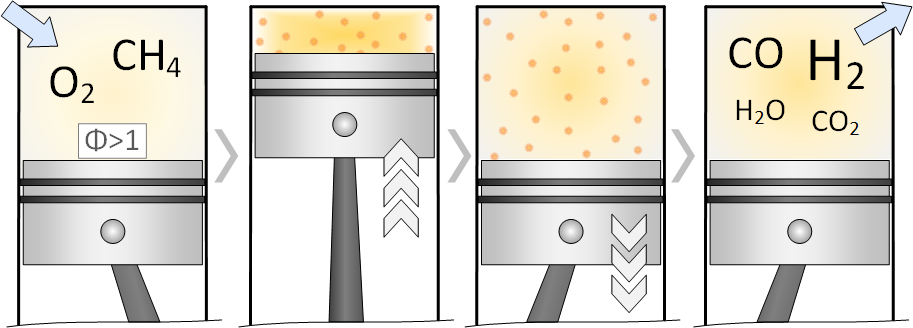
The fuel rich partial oxidation of methane in the HCCI engine model leads to high synthesis gas (CO + H2) yields.
The results show that reactive additives like dimethyl ether, diethyl ether and n-heptane are necessary to achieve practicable low temperatures at the inlet of the engine [1,2]. However, the additives contribute to a high extent to the supplied exergy of the process. A promising, currently investigated alternative is ozone, which can be produced directly from ambient air. As ozone decomposes at low temperatures (200°C), significantly less amounts are needed to achieve the same effect as the additives based on hydrocarbons (0,1 vol-% compared with up to 15 vol-%). The ozone decomposition leads to an earlier formation of radicals which then lead to an earlier and higher conversion of the fuel.
The exergetic efficiency of the engine process reaches up to 81% [2]. If separation processes for the products are considered as well, the efficiency is about 60% [3]. It is then comparable to the efficiency of conventional production processes (steam reforming) but offers a 17%-points higher value compared to the cogeneration of heat and power. Furthermore, the system’s output can be adjusted more flexible according to the demand of work and chemicals.
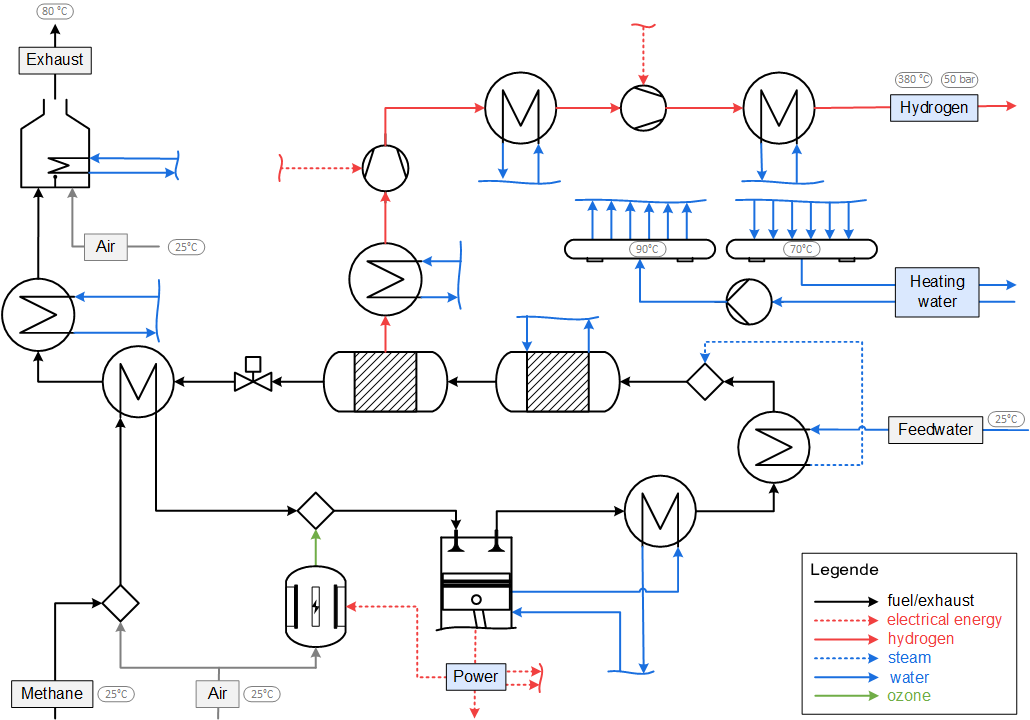
Example of a process flow diagram that is used for the exergoeconomic analysis.
The costs of the hydrogen production for a mid-scale system (engine: 22.6 l displacement) are evaluated at 2 €/kgH2. In comparison with conventional hydrogen production processes (steam reforming) this polygeneration concept is competitive and may be an important contributor to the reliable and efficient energy- and material supply of the near future.