Research Areas
Our work spans HIV persistence, inflammatory host responses, antiviral discovery, immunometabolic virology, and the cell biology of emerging RNA viruses.
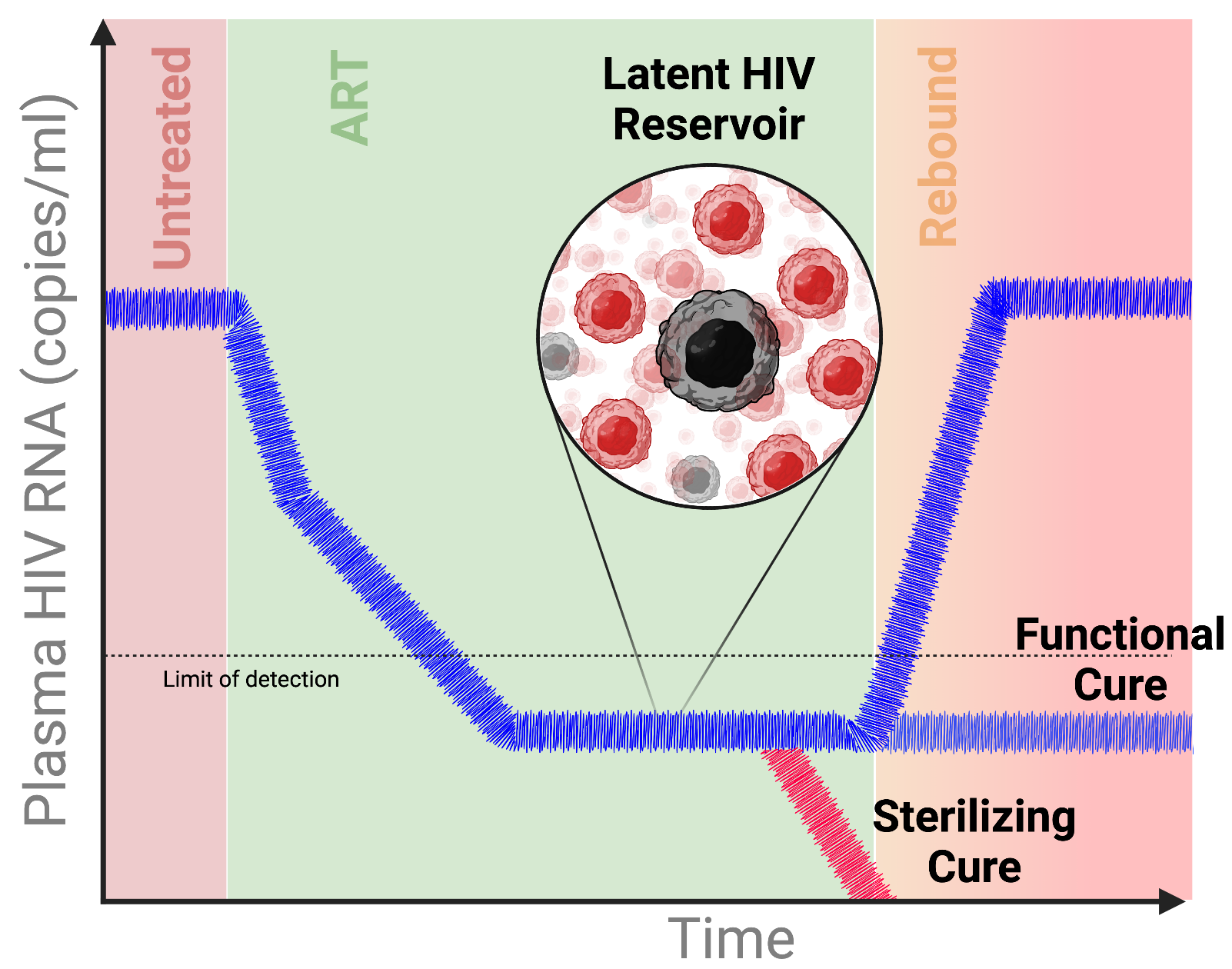
HIV Reservoir and Cure Research
ART transformed HIV into a manageable condition, but it does not eliminate the long-lived reservoir of latently infected cells. These cells persist despite therapy and can reignite infection after treatment interruption. Our research investigates how intrinsic cellular states and tissue-like microenvironments shape HIV latency, persistence, and reactivation. We combine mechanistic virology with patient-derived systems, CRISPR-based perturbation, and functional profiling to identify pathways that stabilize or destabilize the reservoir. The long-term goal is to define actionable vulnerabilities that can be exploited in future HIV cure strategies.

Inflammatory Circuits and Pyroptosis
Abortive HIV infection in tissue CD4⁺ T cells can trigger inflammasome activation and caspase-1-mediated pyroptosis, amplifying local inflammation and immune damage. We study how host factors, tissue signals, and infection context determine whether cells survive, die, or contribute to inflammatory amplification. Using tissue models and mechanistic perturbation approaches, we dissect the pathways that connect viral sensing to immune dysfunction. This work aims to identify intervention points that could reduce inflammatory damage while preserving antiviral control.

Antivirals Discovery
We study antiviral strategies across different viral systems, with a focus on mechanism, delivery, and context-dependent efficacy. This includes both virus-specific and broader host-directed approaches. Our work examines how cellular state, tissue-like conditions, and viral diversity shape therapeutic performance. By combining collaborative screens with mechanistic follow-up, we identify candidates worth advancing and define why others fail. This research connects fundamental virology to translational antiviral development.
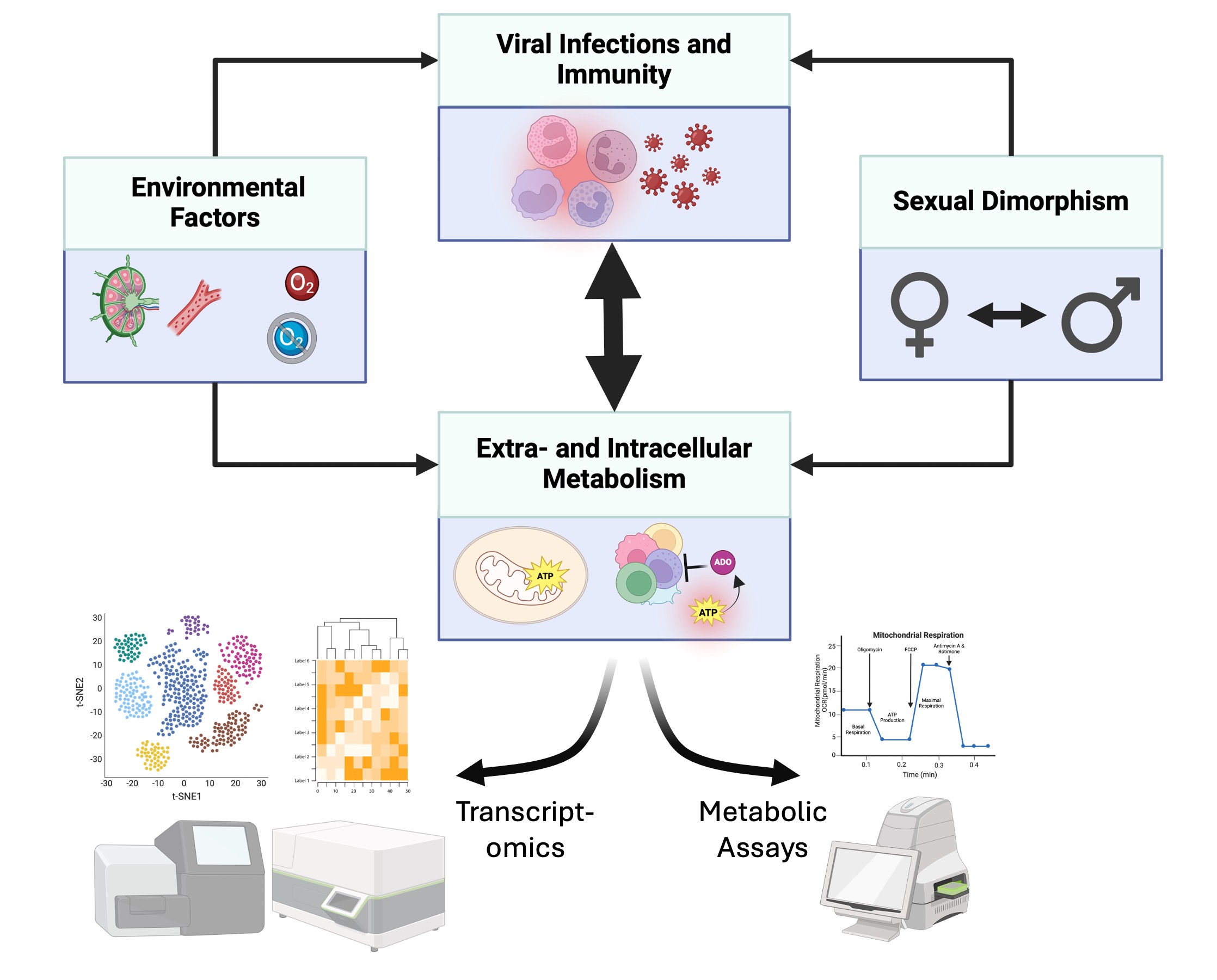
Immunometabolic Virology
Viruses reprogram cellular metabolism, while host metabolic state and the surrounding microenvironment shape infection outcome. We use single-cell profiling, Seahorse and mass spectrometry-based metabolic analysis, and CRISPR perturbation to map metabolic dependencies and identify therapeutic targets. Our aim is to define how oxygen availability, nutrient conditions, immune activation, and metabolic circuits influence whether infection is controlled, amplified, or rendered persistent. This work connects immunology, metabolism, and virology in a mechanistically grounded framework.
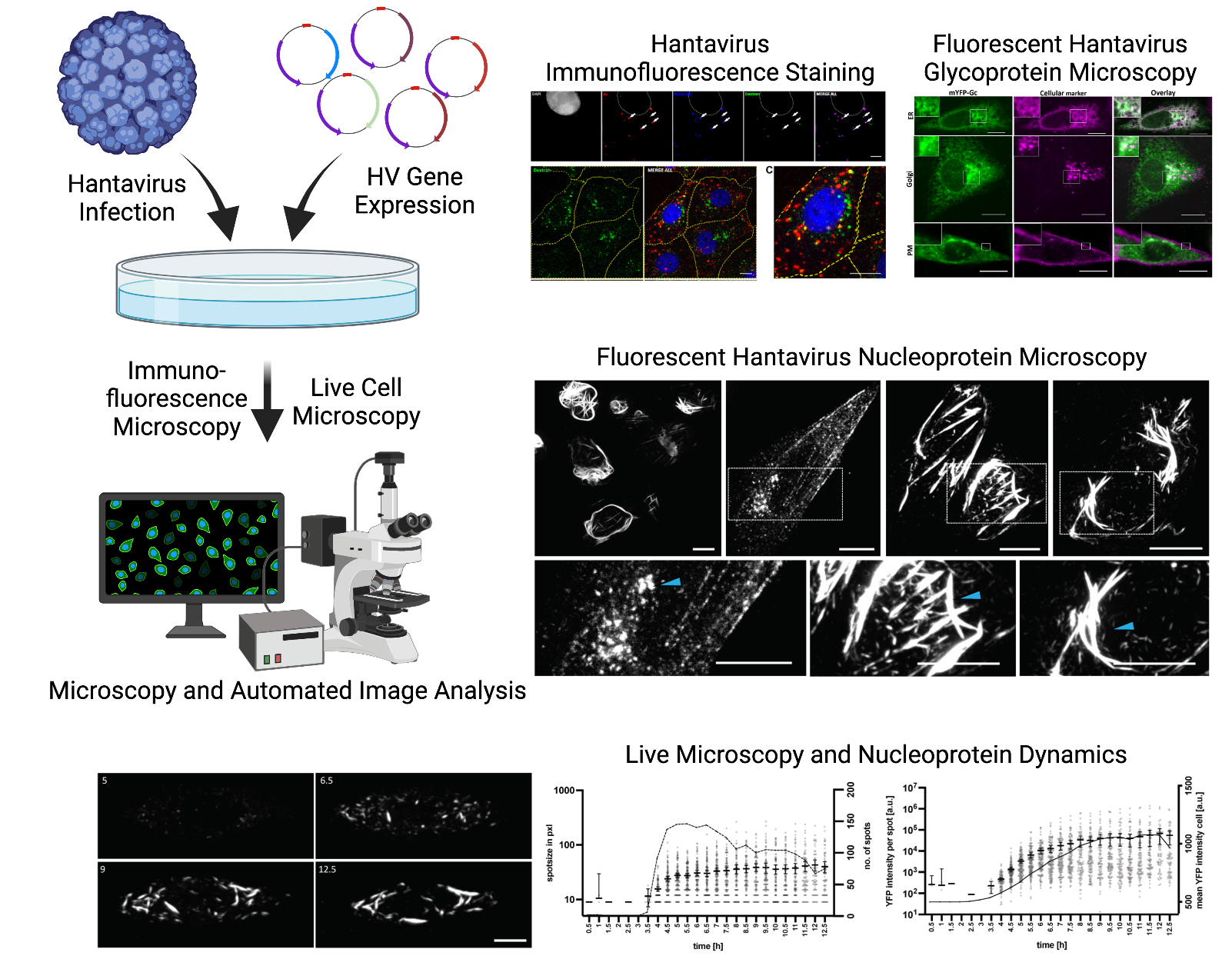
Cytoskeleton and Orthohantavirus Dynamics
Orthohantaviruses are zoonotic, tri-segmented negative-sense RNA viruses that can cause severe human disease. We investigate how viral entry, glycoprotein behavior, cytoskeletal remodeling, and RNA-regulatory compartments such as P-bodies shape infection outcome. Using interactomics, CRISPR, and quantitative microscopy, we map host–virus interfaces including glycoprotein entry pathways, cytoskeletal dependencies, and infection-induced cellular remodeling. By resolving these interactions, we aim to define mechanisms of pathogenesis and identify host-directed antiviral targets.

