Adsorber or catalytic materials
Content
- Design of biopolymer-based adsorbers for water purification
- Environmentally friendly method for synthesis of oxygen reduction catalysts using natural precursors
- Polymer/carbon microspheres for functional nanoparticle-doped composites
- Biopolymer fibers as sustainable media for the removal of micropollutants from wastewater
Design of biopolymer-based adsorbers for water purification
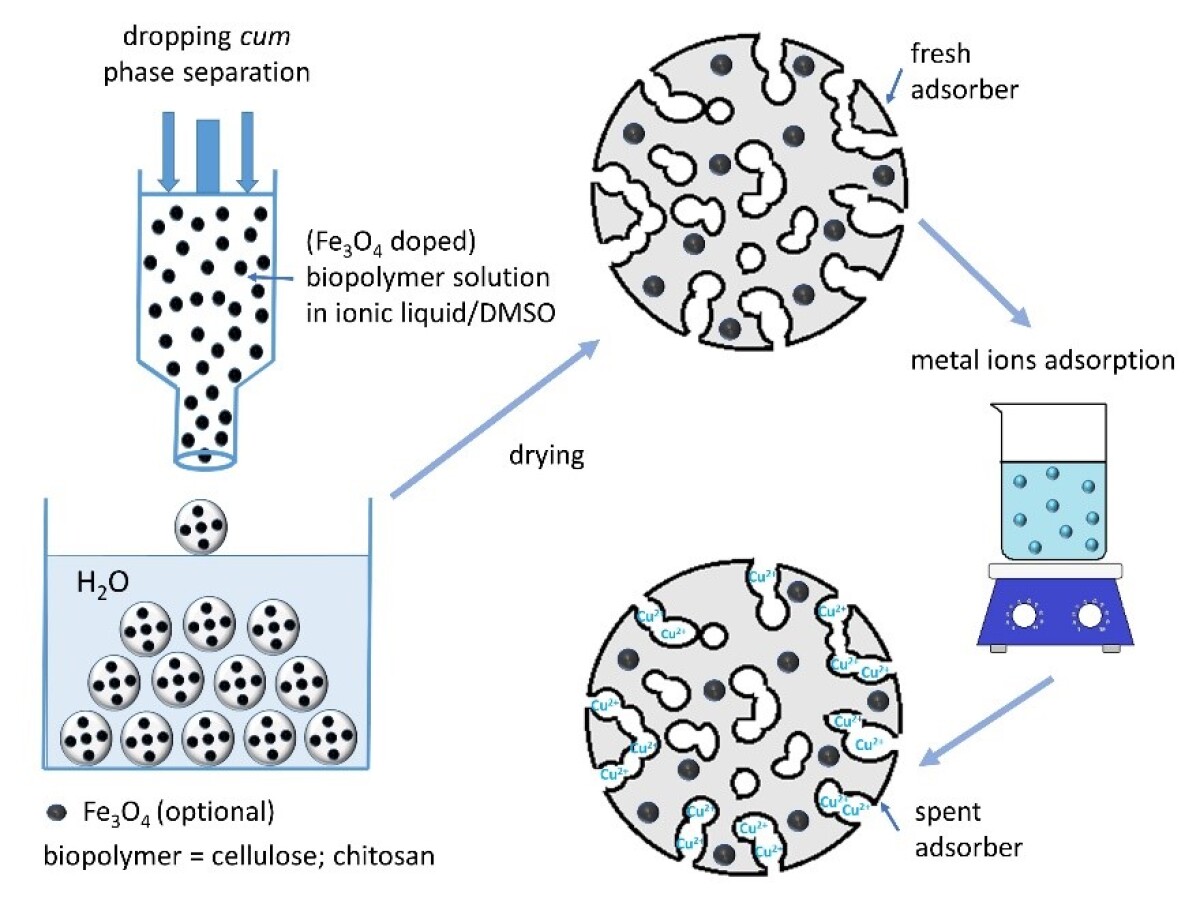
The continuous increase of world population and the industrialization have led to an increased demand for fresh clean water. Of very high interest is the development of new and efficient adsorbers, based on renewable and biodegradable biopolymers, for the removal of different pollutants such as toxic heavy metals, dyes, fertilizers, or phenolic compounds from water. The materials or processing aids used in the project should be mainly based on renewable biopolymers (cellulose, chitosan), well-established functional nanoparticles (e.g. Fe3O4) and environmentally friendly solvents (ionic liquids, water).
In this project, research is focused on establishing a platform for the direct processing of the biopolymers from their solutions in ionic liquids (optionally containing a fraction of another solvent, without or with dispersed nanoparticles) towards porous adsorber particles of spherical shape with defined size ≤ 1 mm along with adjustable pore size distribution and high specific surface area. The functionality of the porous spheres is provided by blending cellulose with chitosan (adsorber groups) and by doping with functional nanoparticles (magnetic properties). The spheres in mm range are prepared by dropping cum phase separation technique while smaller spheres are prepared by emulsion techniques. The smaller spheres will be prepared only for comparison reasons. The influence of different parameters like spheres size, pore structure and chitosan content on the adsorption properties against different metal ions (copper, manganese, zinc) is investigated in detail using series of porous cellulose/chitosan beads prepared by non-solvent induced phase separation (NIPS) techniques. The selectivity of the adsorption towards certain metal ion species is also evaluated.
References:
[1] A. S. M. Wittmar, J. Klug, M. Ulbricht, Carbohydrate Polymers, 2020, 237, 116135.
[2] A. S. M. Wittmar, H. Böhler, A. L. Kayali, M. Ulbricht, Cellulose, 2020.
Contact: Dr. Alexandra Wittmar
Environmentally friendly method for synthesis of oxygen reduction catalysts using natural precursors
The current objective to maintain global warming below 2 °C above preindustrial levels is strongly connected with the reduction of emissions of greenhouse gases, especially of CO2. This can be achieved when greener and more sustainable energy technologies such as water splitting for hydrogen production, fuel cells or metal air batteries are further developed and improved. All these technologies are based on two chemical reactions that affect the efficiency of energy conversion and storage: the oxygen reduction reaction (ORR) and the oxygen evolution reaction (OER).
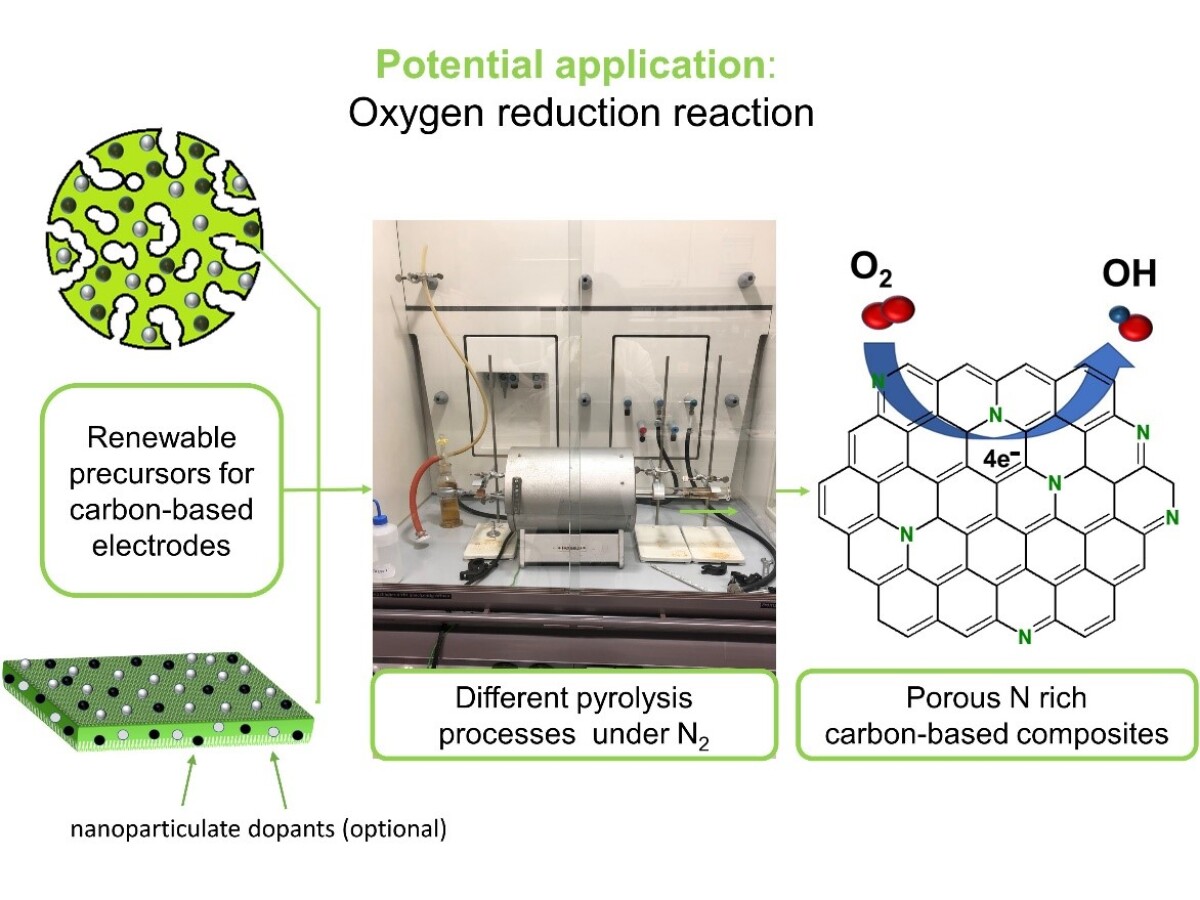
In the present work we focus on the development of a procedure which allows the conversion of environmentally friendly and renewable biopolymers cellulose and chitosan to nitrogen-doped carbon materials with good catalytic activity in the ORR. We aim to perform the pyrolysis processes in such a manner as to minimize the use of additives and to reduce the emissions to the atmosphere of corrosive toxic gases, and by this to minimize the impact on the environment. Porous cellulose and cellulose/chitosan blends with different compositions are prepared by shaping from solutions and phase separation and subsequently used as precursors for the pyrolysis process. Nitrogen rich additives like ammonia or melamine may be used in combination with the biopolymers in order to increase the nitrogen content in the final pyrolyzed product, while other additives may be used in order to increase the specific surface area of the materials. The aim is to establish the optimal pyrolysis conditions, in terms of precursors composition, heating rate, maximal temperature and pyrolysis duration, which will allow the synthesis of nitrogen doped carbons with high number of graphitic and pyrrolic sites along with optimized pore structure and thus high catalytic activity.
Contact: Dr. Alexandra Wittmar
Polymer/carbon microspheres for functional nanoparticle-doped composites
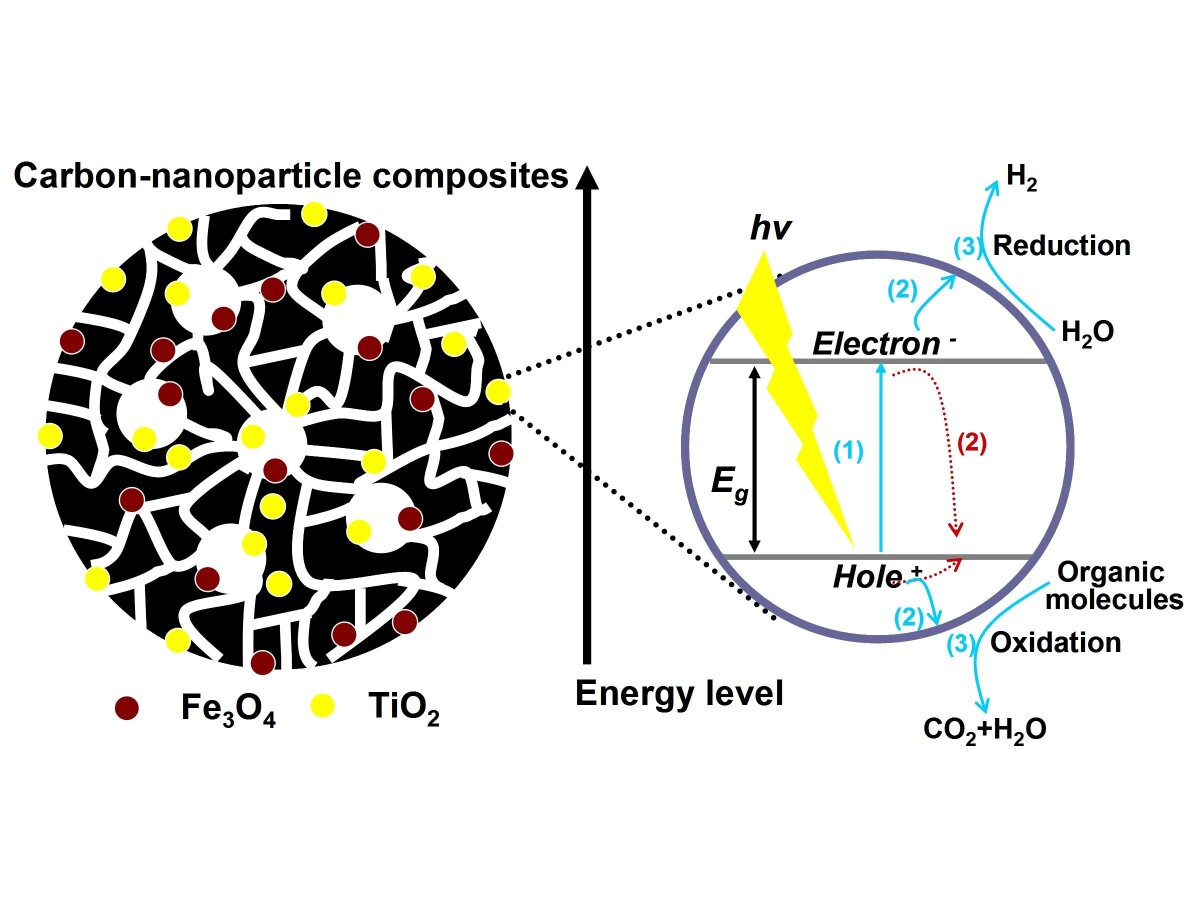
Nanoscale inorganic particles, such as TiO2, are popular in the field of photocatalysis like water splitting reaction or pollutant degradation [1, 2]. However, due to the aggregation effects and ultra-small size of these nanoparticles, increasing the operability and recoverability is a vital issue for various applications. Porous polymer or carbon spheres as carriers for the nanoparticles can overcome these problems. This project aims to prepare nanocomposites by combining functional nanoparticles and porous polymer or carbon spheres in a simple blending way. Furthermore, porous microspheres can be easily recycled when adding certain magnetic materials, such Fe3O4.
Polyacrylonitrile (PAN) is selected as precursor and can be efficiently pyrolyzed into nitrogen containing carbon materials. Porous polymer spheres with diameter in the µm-range are prepared by microdroplet shaping (similar to ink-jet printing) cum non-solvent induced phase separation (NIPS) [3], starting from PAN solutions. Polymer nanocomposites as well as carbon nanocomposites resulted from carbonization are used for adsorption and photocatalysis characterization. In addition, the nitrogen containing carbon materials are also evaluated electrocatalysts. Pure PAN spheres after NIPS have a large surface area (250 m2/g), and this value decreased drastically almost to zero after pyrolysis. However, this problem can be solved by carbonizing the carbon spheres under CO2 and N2 atmosphere. Shaping intact PAN microspheres with diameter of about 50 µm is also possible if detailed adjustment of molecular weight, concentration, and temperature of the polymer solution is done. Decreasing the driving voltage and shaping frequency of the ink-jet printer-like device as well as increasing the height above the non-solvent coagulation bath are an effective way to acquire perfect microspheres. Combining functional nanoparticles and polymer into polymer- or carbon-based composites as well as corresponding catalytic characterization are still undergoing.
References:
[1] H. Thierfeld, Herstellung poröser Polymer- und Kohlenstoff-basierter Hybridmaterialien zur Adsorption und Photokatalyse, Dissertation, Universität Duisburg-Essen, 2018.
[2] A. Wittmar, Q. Fu, M. Ulbricht, ACS Sustainable Chemistry & Engineering, 2017, 5, 9858-9868.
[3] M. Ulbricht, Polymer, 2006, 47, 2217-2262.
Funding: The financial support from Chinese Scholarship Council is kindly acknowledged.
Contact: Zhuo Gao, M.Sc.
Biopolymer fibers as sustainable media for the removal of micropollutants from wastewater
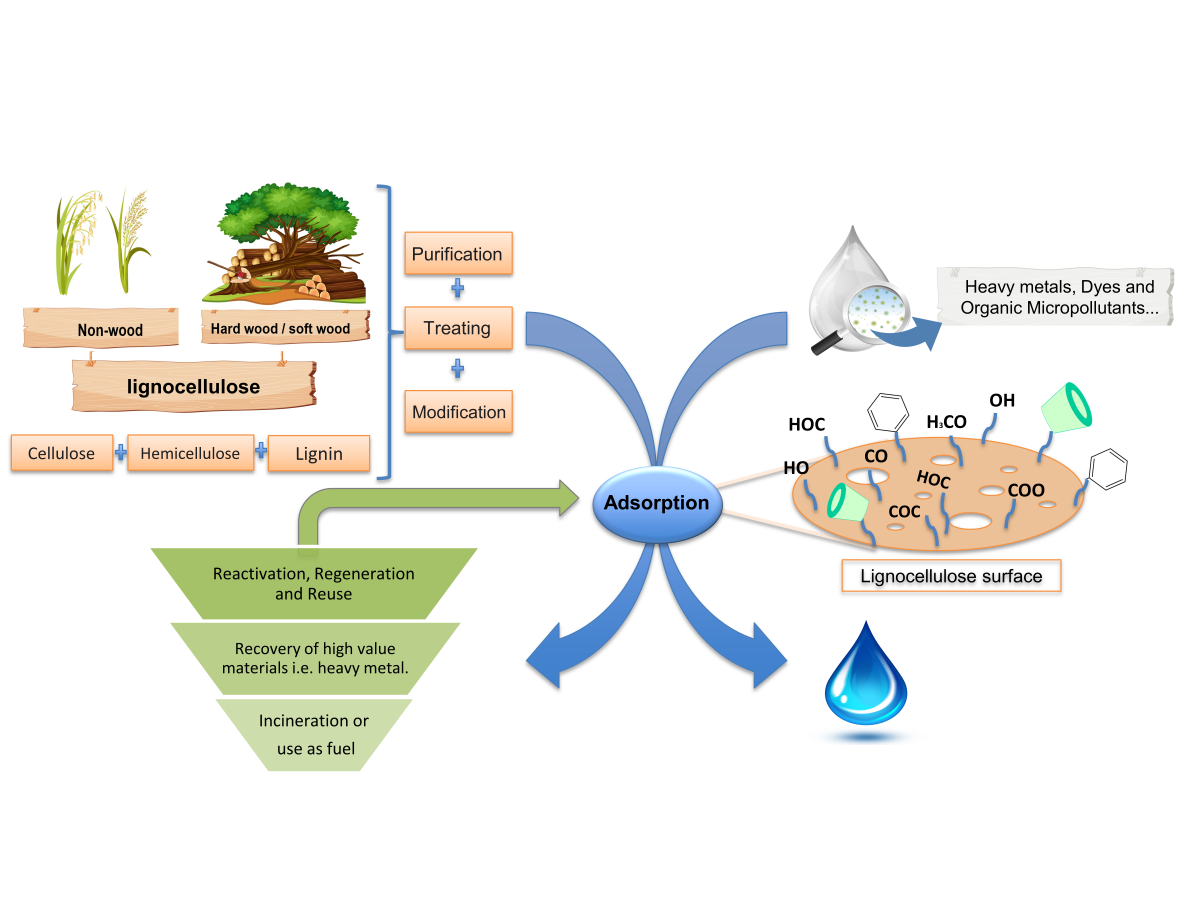
Chemical pollutants, i.e., heavy metals, dyes, fluorinated surfactants, as well as other micropollutants such as endocrine disruptors, hormones and antibiotics, pose a massive threat to human life and the environment. Adsorption is one of the most favorable water treatment methods due to its high treating impact, convenience, simplicity of design as well as low side effects and environmental impact. Lignocellulose, a renewable, sustainable, and low-cost biomaterial, shows high potential for removing pollutants from water by adsorption. Lignocellulose is a complex polymer made up of three main components, cellulose, hemicellulose, and lignin, with minor amounts of extractives. It can be obtained from different sources such as hardwood, softwood, and non-wood plants. The structure and chemical composition of the lignocellulose varies depending on the source, and these changes have a significant influence on the adsorption process. Furthermore, the purification and the modification of lignocellulose could improve its adsorption properties.
This project aims to investigate the possibility of using lignocellulosic fibers from different sources as bio-adsorbent materials to remove various pollutants from wastewater. The project also covers studying the possible treatment of lignocellulose fibers and the potential structure modification that could enhance the target pollutants' removal efficiency. The effect of the types and density of functional groups, surface area, porosity, hydrophilicity, and overall surface charge, which vary according to the lignocellulose source and the modification process, are investigated. Further work focuses on determining the optimal adsorption operational parameters. Studies of the effect of shaping lignocellulosic fibers in different forms, including spherical shape granules and membranes, on the efficiency of wastewater treatment will also be included in this research work.
Funding: This project is funded by the DAAD (Deutscher Akademischer Austauschdienst).
Contact: Mhd Mazen Altayan, M.Sc.