Research
Research
Chemical Biology to Unravel Protein Homeostasis
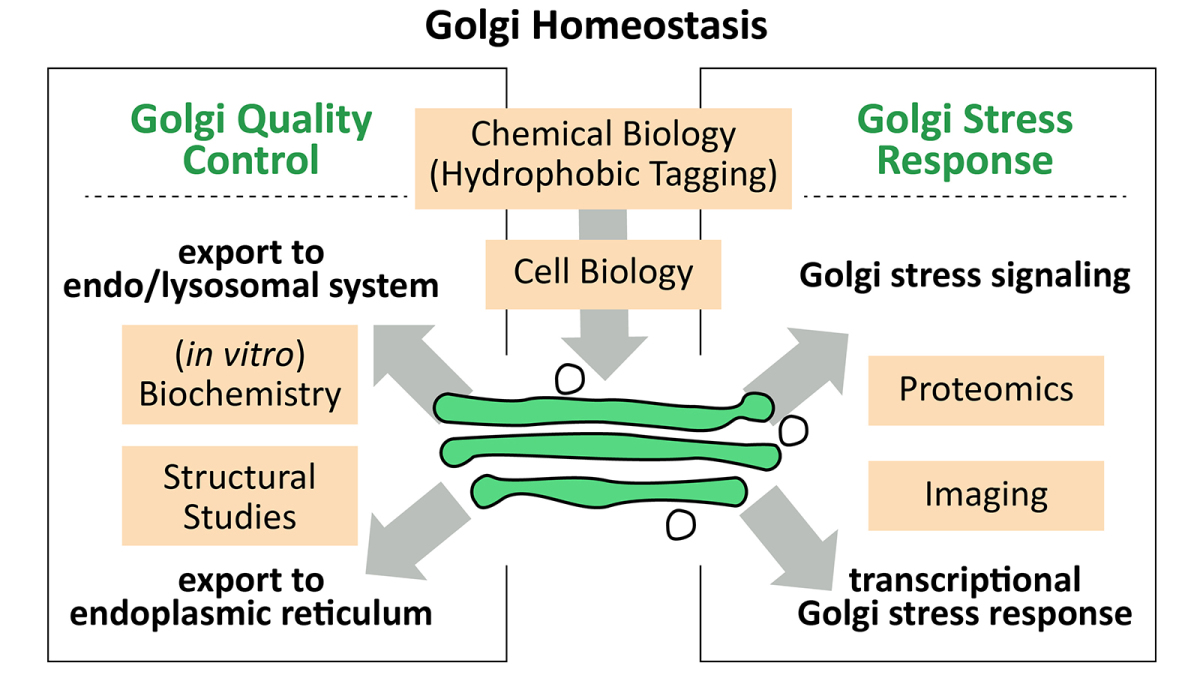
Understanding how cells maintain homeostasis is critical to understanding cell survival during stress. Cells have to withstand stress from changing environmental conditions, such as temperature, pH, nutrient or oxygen supply, but also during cell differentiation and aging. Eukaryotic cells have evolved to use distinct cellular compartments, known as organelles, which maintain homeostatic mechanisms to ensure their distinct functionalities and overall cellular homeostasis. Within the secretory pathway, these stress response mechanisms have been extensively studied at the level of the endoplasmic reticulum (ER). In contrast, the mechanism(s) by which the Golgi apparatus deals with unfolded proteins and how it reacts to stress are only beginning to be unraveled.
Studying and identifying protein quality control (PQC) and stress response pathways relies on model systems that allow inducing organelle-specific stress. To this end, we are using chemical biology tools that enable manipulating the folding state of a model protein. Applying this approach in the Golgi revealed a transcriptional stress response to protein unfolding. We find that PQC substrates are exported from the Golgi; where aggregated proteins are degraded in lysosomes, while unfolded proteins localize to the ER and associate with ER-chaperones.
Future work will focus on identifying molecular mechanisms underlying Golgi stress response and PQC pathways. The combined in vitro and in cellulo efforts are aimed at delineating the Golgi homeostasis network.