Research
The VCP/p97 system and cellular stress responses
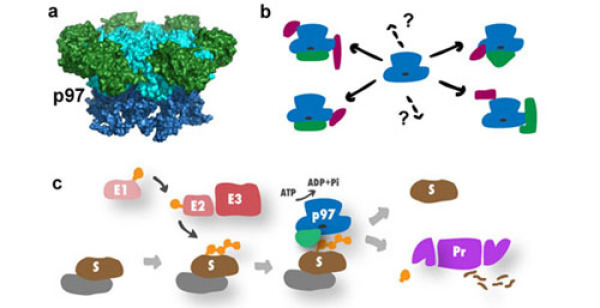
The abundant hexameric AAA+-type ATPase p97 (also called VCP or Cdc48) has emerged as a key regulatory molecule in many ubiquitin-regulated reactions that mediate and coordinate cellular stress responses. It assembles into alternative holo-complexes with diverse ubiquitin adapters and other accessory proteins. By targeting and processing ubiquitinated factors, distinct p97 complexes coordinate key decisions points in diverse pathways. In the past, we have revealed basic principles of how p97 cooperates with ubiquitin adapters such as the Ufd1-Npl4 adapter or accessory factors including the VCPIP1 deubiquitinating enzyme. Moreover, we have linked p97 function to chromatin-related processes and the endolysosomal system. Our current focus is to identify and characterize p97-cofactor complexes, understand their function on the mechanistic level and reveal their role in human with respect to maintenance of cellular homeostasis and cell proliferation. To achieve this, we use a combination of proteomics, biochemical reconstitution, modern cell biological approaches and light microscopy.
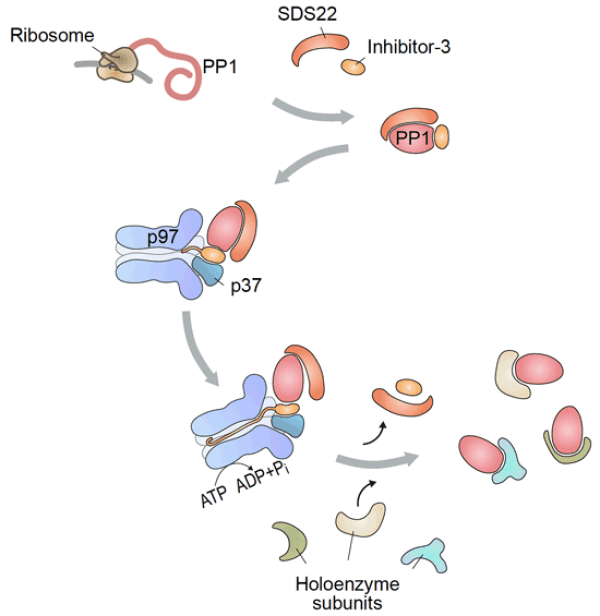
Recent Advances Ubiquitin-independent disassembly by a p97 AAA-ATPase complex drives PP1 holoenzyme formation
Protein phosphatase-1 (PP1) dephosphorylates a broad spectrum of substrates to regulate a myriad of cellular processes. This functional diversity relies on the ordered assembly of alternative PP1 holoenzymes. Here, we show that newly synthesized PP1 is first held in an inactive complex by its partners SDS22 and inhibitor-3 (I3) that needs to be disassembled by the p97 AAA-ATPase to promote exchange to substrate specifiers. Unlike p97-mediated degradative processes that require the Ufd1-Npl4 ubiquitin adapters, p97 is targeted to PP1 by p37 and related adapter proteins. Reconstitution with purified components revealed direct interaction of the p37 SEP domain with I3 without the need of ubiquitination, and ATP-driven pulling of I3 into the central channel of the p97 hexamer, which triggers dissociation of I3 and SDS22. Thus, we establish regulatory ubiquitin-independent protein complex disassembly as part of the functional arsenal of p97 and define an unanticipated essential step in PP1 biogenesis that illustrates the molecular challenges of ordered subunit exchange.
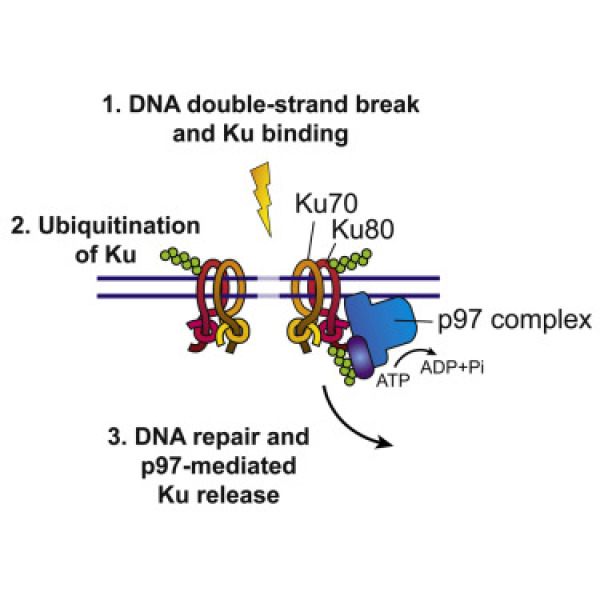
VCP/p97 extracts sterically trapped Ku70/80 rings from DNA in double strand break repair Research-hemmo-meyer-figure2
DNA double strand breaks (DSBs) is the most severe type of DNA damage, which can lead to cell death or genomic instability. Cells have developed sophisticated mechanisms to repair the damage and coordinate that with cell cycle progression. During repair of DSB, the ring-shaped Ku70/80 complex becomes trapped on DNA and needs to be actively extracted. By means of in vitro reconstitution of DSB repair on beads, we have demonstrated that DNA-locked Ku rings are released by the AAA-ATPase, p97. To achieve this, p97 requires ATP hydrolysis, cooperates with the Ufd1-Npl4 ubiquitin adapter complex and specifically targets Ku80 that is modified by K48-linked ubiquitin chains. We also showed that, in U2OS cells, chemical inhibition of p97, or siRNA-mediated depletion of p97 or its adapters impairs Ku80 removal after non-homologous end-joining of DSBs. Moreover, p97 inhibition attenuates early steps in homologous recombination consistent with p97-driven Ku release also affecting repair pathway choice. Thus, our data solve a central question regarding regulation of Ku in DSB repair, and illustrate the ability of p97 to segregate even tightly bound protein complexes for release from DNA.
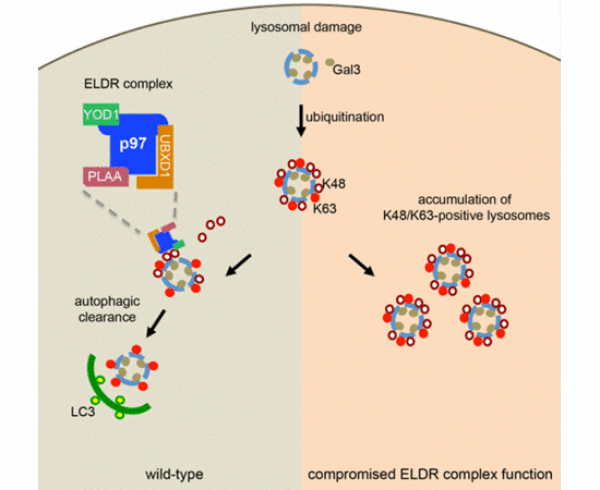
VCP/p97 cooperates with the cofactors YOD1, UBXD1 and PLAA to drive clearance of ruptured lysosomes by autophagy
Rupture of endosomes and lysosomes is a major cellular stress condition leading to cell death and degeneration. We have identified an essential role for the ubiquitin‐directed AAA‐ATPase, p97, in the clearance of damaged lysosomes by autophagy. Upon damage, p97 translocates to lysosomes and there cooperates with a distinct set of cofactors including UBXD1, PLAA, and the deubiquitinating enzyme YOD1, which we term the ELDR complex for Endo‐Lysosomal Damage Response. Together, they act downstream of K63‐linked ubiquitination and p62 recruitment, and selectively remove K48‐linked ubiquitin conjugates from a subpopulation of damaged lysosomes to promote autophagosome formation. Lysosomal clearance is also compromised in MEFs harboring a p97 mutation that causes inclusion body myopathy and neurodegeneration, and damaged lysosomes accumulate in affected patient tissue carrying the mutation. Moreover, we show that p97 helps clear late endosomes/lysosomes ruptured by endocytosed tau fibrils. Thus, our data reveal an important mechanism of how p97 maintains lysosomal homeostasis, and implicate the pathway as a modulator of degenerative diseases.