Carbon
Graphene and C60 films
Very thin graphite layers (graphene) have remarkable electronic properties. They promise to be high-mobility conductors and may be doped by small amounts of impurities. On SiC surfaces such layers may be prepared by thermal decomposition. If undoped, they show a semi-metal bahaviour. Exposure to small amounts of Cs atoms metallizes the C layers. This is demonstrated in Fig. 1 where the work function is plotted as a function of the Cs exposure. The sudden decrease of the work function by 1.8 eV indicates that valence electrons of Cs atoms is transferred to the graphene layer.
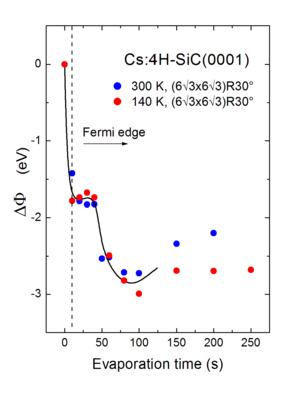
Fig 1: The change of workfunction of a graphene layer on SiC as a function of the Cs exposure. The rapid decrease even after very small doses indicates the doping of the carbon layer.
An impressive example of the chemical passivation of surfaces by carbon is the oxidation of the technological important wide-bandgap semiconductor GaN. GaN surfaces are easily oxidized by ambient air. This oxidation is promoted at step edges. If the surface and especially the steps are covered by C60 molecules the oxidation process is suppressed by orders of magnitude. Fig. 2 shows schematically a C60 molecule on a GaN step and in Fig. 3 the oxygen uptake of clean and carbon covered GaN surfaces is plotted as a function of the oxygen exposure on a logarithmic scale!
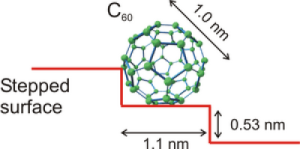
Fig 2.: Schematics of a C60 on a GaN surface step.
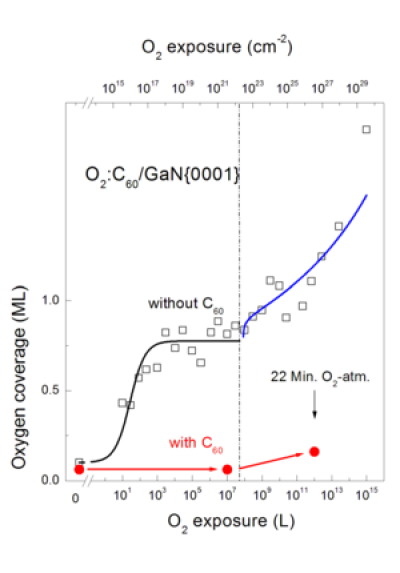
Fig 3: C60 layers on GaN prevent surface oxidation efficiently. The oxygen coverages of clean and C60 covered GaN surfaces are shown as a function of the oxygen exposure.
