Research
Iterative Methods for Organic Synthesis
In our vision the ultimate synthetic method is one that allows for the assembly of individual C1-buildingblocks with perfect control over hybridization, regio- / stereoselectivity, annular connectivity, as well as heteroatom-substituents. If such a hypothetical method could be automated a piece of “Star Trek” technology would be realized: the replicator, which assembles matter atom by atom.
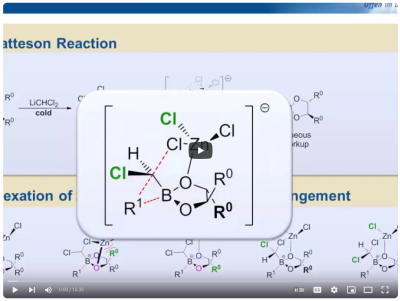
Although such a machine is still science fiction, we are convinced that the underlying concept has the potential for important innovations in the here and now. Currently the most powerful methods for iterative C1-homologations are based on 1,2-metallate rearrangements of boron-carbenoid complexes. The first such process was developed by D. Matteson in the last two decades of the 20th century and it allows for the assembly of substituted alkyl chains in a similar way, as one would build a molecular model.

Modern variants mostly use chiral carbenoids, which bring several advantages (reagent control, insertion of quarternary stereocenters etc.). However, the Matteson-Homologation (MH) also has its merits and is especially useful for the synthesis of heteroatom-rich motives. We contribute to the field by further developing these methods and applying them in the synthesis of important and challenging targets.
Synthesis of Natural Products and Bioactive Compounds
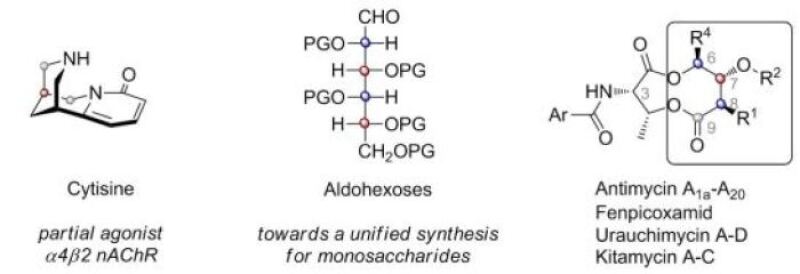
As targets for synthesis we like to choose natural products not only because of their potential applications, but also because of the specific challenges they pose to iterative synthesis. This includes bicyclic molecules such as Cytisine (video-abstract) or heteroatom-rich molecules like monosaccharides and certain macrolactones (video-tutorial). These motives challenge current methodology, which can deal well with EITHER differentially protected heteroatoms OR repeating threo-connections (video abstract)
Methodology
Thus our work on natural products is complemented by further development of homologation methodology. Two underlying concepts are shown below.
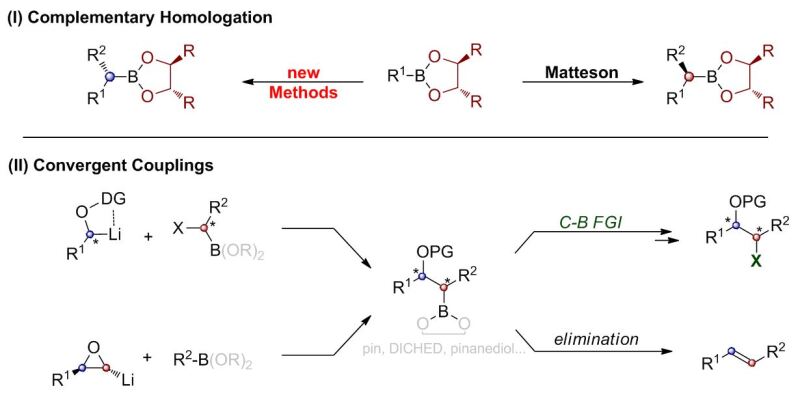
(I) Complementary Homologation Methodology
MH is a great tool for assembling highly substituted alkyl chains that contain multiple heteroatom substituents with individual protecting groups in an erythro-relationship. However, current methods for preparing threo-configurations are limited. They usually require a change of chiral director, which can be done efficiently, only once (by transesterification from DICHED to pinanediol: video-abstract). Therefore we are looking for complementary methods for MH that assigns a new mechanistic role to the chiral director.
(II) Convergent Couplings
Every linear synthesis that doesn´t enjoy reliably quantitative yields suffers from significant loss of material over multi step syntheses. Thus we are looking at the development of methods that allow for the connection of two complementary carbon chains in order to make differentially protected 1,2-systems (video-abstract). Here a key challenge is to either control for competing elimination reactions, or harnesses them in a desired fashion (video-abstract). This e.g. led us to the development of an epoxide olefination, which can be applied in an iterative manner (video-abstract).
Relevant Literature:
Boron-Carbenoid Homologations in Context: (a) S. Kirupakaran, H.-G. Korth, C. Hirschhäuser, Synthesis 2018, 50, 2307-2322; (b) S. P.Thomas, R. M. French, V. Jheengut, V. K. Aggarwal The Chemical Record 2009, 9, 24-39.
Matteson-Homologations: Review: (a) D. S. Matteson J. Org. Chem. 2013, 78, 10009-10023; Original Articles: (b) D. S. Matteson, D. Majumdar J. Am. Chem. Soc. 1980, 102, 7588-7590; (c) D. S. Matteson, R. Ray J. Am. Chem. Soc. 1980, 102, 7590-7591.
Homologations with Chiral Carbenoids: (a) D. Leonori, V. K. Aggarwal, Acc. Chem. Res. 2014, 47, 3174-3183; (b) H. K. Scott, V. K. Aggarwal, V. K. Chem. Eur. J. 2011, 17, 13124-13132; (c) B. S. L. Collins, C. M. Wilson, E. L. Myers, V. K. Aggarwal, Angew. Chem. Int. Ed. 2017, 56, 11700-11733; (d) F. Pape, J. F. Teichert, Nachr. Chem. 2017, 65, 879-883.
Synthesis of Cytisine: (a) C. Hirschhäuser, C. A. Haseler, T. Gallagher, Angew. Chem. Int. Ed. 2011, 50, 5162–516; (b) F. R. Struth, C. Hirschhäuser, Eur. J. Org. Chem. 2016, 2016, 958–964; Review on earlier syntheses: (c) D. Stead, P. O´Brien Tetrahedron, 2007, 63, 1885-1897.
Diols from Carbenoids: (a) D. S. Matteson, P. B. Tripathy, A. Sarkar, K. M. Sadhu, J. Am. Chem. Soc. 1989, 111, 4399–4402.
Akenes from Carbenoids: (a) K. Bojaryn, C. Hirschhäuser Chem. Eur. J. 2022, 28, e202104125 ; (b) P. R. Blakemore, R. W. Hoffmann, Angew. Chem. Int. Ed. 2018, 57, 390–407. Review on lithiated epoxides: (c) D. M. Hodgson, E. Gras, Synthesis 2002, 2002, 1625–1642; (d) V. Capriati, S. Florio, R. Luisi, Chem. Rev. 2008, 108, 1918–1942; Also see SI of: (e) K. Bojaryn, S. Fritsch, C. Hirschhäuser, Org. Lett. 2019, 21, 2218-2222.
Diols from Epoxides: (a) E. Vedrenne, O. A. Wallner, M. Vitale, F. Schmidt, V. K. Aggarwal, Org. Lett. 2009, 11, 165-168; (b) R. J. Armstrong, V. K. Aggarwal, Org. Synth. 2017, 94, 234-251; (c) E. Alwedi, L. N. Zakharov, P. R. Blakemore, Eur. J. Org. Chem. 2014, 2014, 6643–6648; (d) K. Bojaryn, C. Hoffmann, F. R. Struth, C. Hirschhäuser, Synlett 2018, 29, 1092–1094.
C-B FGIs: a) C. Sandford, V. K. Aggarwal, Chem. Commun. 2017, 53, 5481-5494.
Alkenes from Epoxides: (a) M. Shimizu, T. Fujimoto, H. Minezaki, T. Hata, T. Hiyama, J. Am. Chem. Soc. 2001, 123, 6947–6948; (b) K. Bojaryn, S. Fritsch, C. Hirschhäuser, Org. Lett. 2019, 21, 2218-2222; (c) S. Fritsch, N. Aldemir, J. Balszuweit, K. Bojaryn, J. Voskuhl, C. Hirschhäuser, ChemistryOpen 2022, 11, e202200098.