Plasma Synthesis for Novel Materials
Microwave plasma reactor
A plasma can be called the fourth state of matter beside the solid, liquid and gaseous state. It consists out of molecules, atoms, ions, electrons and photons and can reach high temperatures and energy densities. The microwave plasma reactors we use operate in a temperature range of a few thousand Kelvin and thus enable similar operating conditions in terms of the temperature-time profile to those found in flame reactors. In contrast to these, however, neither fuels nor oxidizers are required, so that reaction conditions under inert or reducing atmospheres are also possible. Thus, the microwave plasma reactor allows to synthesize a wide range of nanomaterials, however, so far it requires vaporizable precursors.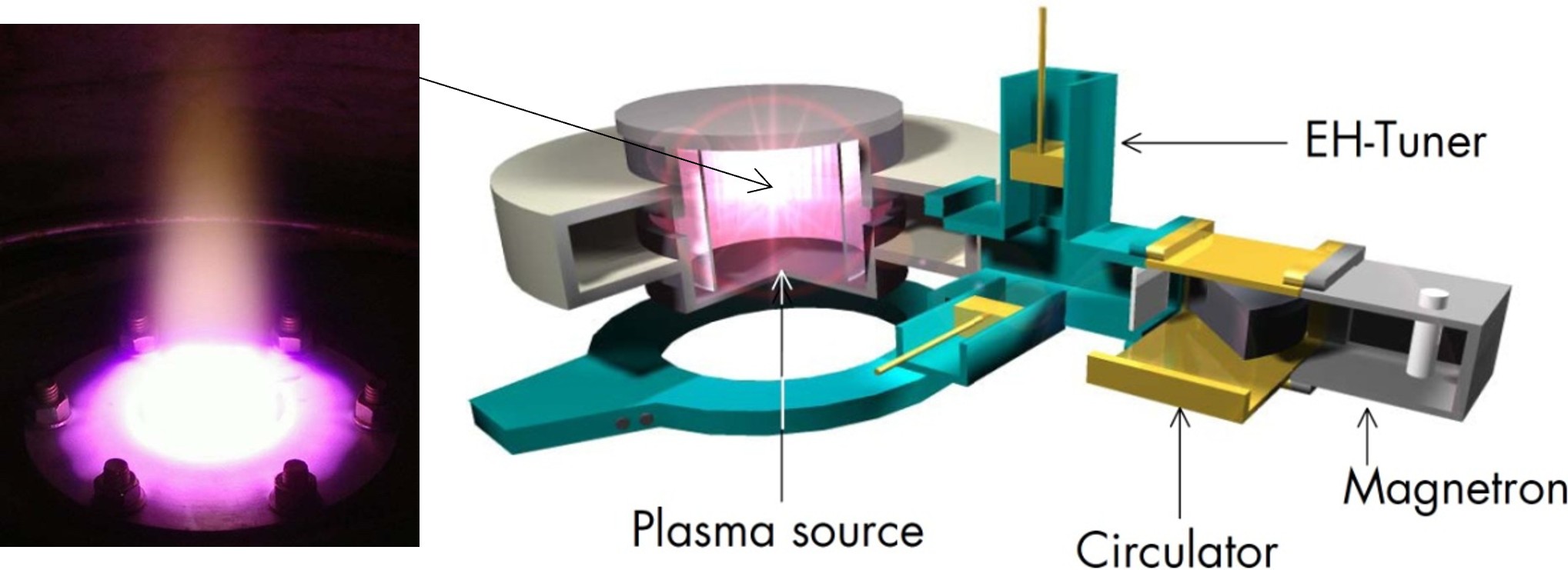
Figure: Left: Afterglow of a microwave plasma reactor (purple to white) and beam of silicon nanoparticles (yellow, flame-like structure). Right: Schematics of the iplas microwave plasma source
Literature:
[1] N. Petermann, T. Schneider, J. Stötzel, N. Stein, C. Weise, I. Wlokas, G. Schierning, H. Wiggers, Microwave plasma synthesis of Si/Ge and Si/WSi2 nanoparticles for thermoelectric applications, J. Phys. D: Appl. Phys. 48 (2015) 314010, DOI: https://doi.org/10.1088/0022-3727/48/31/314010
[2] C. Tendero, C. Tixier, P. Tristant, J. Desmaison, P. Leprince, Atmospheric pressure plasmas: A review, Spectrochimica, Acta Part B: Atomic Spectroscopy 61 (2006) 2–30, DOI: https://doi.org/10.1016/j.sab.2005.10.003.
Radio frequency plasma reactor
Compared to our microwave plasma reactors, radio frequency (RF) plasma reactors produce thermal plasmas with maximum temperatures of more than 10.000 K. Thermal plasmas have a wide range of applications as they are able to vaporize numerous materials due to the high plasma temperatures. They can therefore be used for the production of nanomaterials in a highly pure and controlled atmosphere via evaporation and condensation. RF plasmas are inductively-coupled plasmas (ICP). Due to the fact that all kinds of precursors like solid (in form of powders), liquids, suspensions and gases can be used the production of a wide range of materials like metals, alloys, ceramics etc. is possible.
So, in total this process is a combination of a top-down and bottom-up route in the synthesis of nanoparticles. By changing process parameters, the properties like size, composition and structure of these particles can be controlled and a tailor-made product is the result.
Our RF plasma device (TekNano-15, TEKNA) enables the production of nanoscale silicon from microscale metallurgical silicon. During the process (see Figure 1), an electrical power of up to 15 kW and the resulting very high temperatures cause the microscale silicon to completely vaporize, followed by condensation through controlled cooling and nucleation from the gas phase. Nanoscale silicon is produced. Among other things, the thermodynamic and kinetic conditions can be specifically controlled by varying the composition and flow rate of the quench gas as well as the feed rate of the precursor. This allows product properties to be influenced.
Nanoscale silicon can reduce cracking, which can occur due to volume expansion of up to 300% during lithiation, thereby improving electrochemical performance. Compared to microscale silicon, our product exhibits significantly better electrochemical properties and is therefore a promising anode material for lithium-ion batteries.

Figure 2: Schematic representation of the RF plasma reactor.

