Michael Ehrmann

Prof. Dr. Michael Ehrmann (Retired)
Faculty of Biology
Center of Medical Biotechnology
University of Duisburg-Essen
Overview
Mechanism and Translational Aspects of Protein Quality Control
Each cell hosts thousands of different proteins that vary greatly in copy number and in their chemical properties. Despite this complication, nature has evolved efficient mechanisms of quality control to ensure that all proteins are biologically active, localised to the proper cellular compartment and present in appropriate quantity.
This functional state must be maintained under normal as well as under stress conditions. The failure of quality control can influence cell growth and can cause severe diseases ranging for example from bacterial infections to neurodegenerative and arthritic diseases or cancer.
We are studying evolutionarily conserved cellular factors that are involved in key aspects of quality control, such as detection of misfolded proteins, signal recognition and integration into the unfolded protein response pathways and regeneration of the functional state. These studies aim at revealing the general concepts governing the underlying molecular mechanisms of protein diagnosis, repair and degradation.
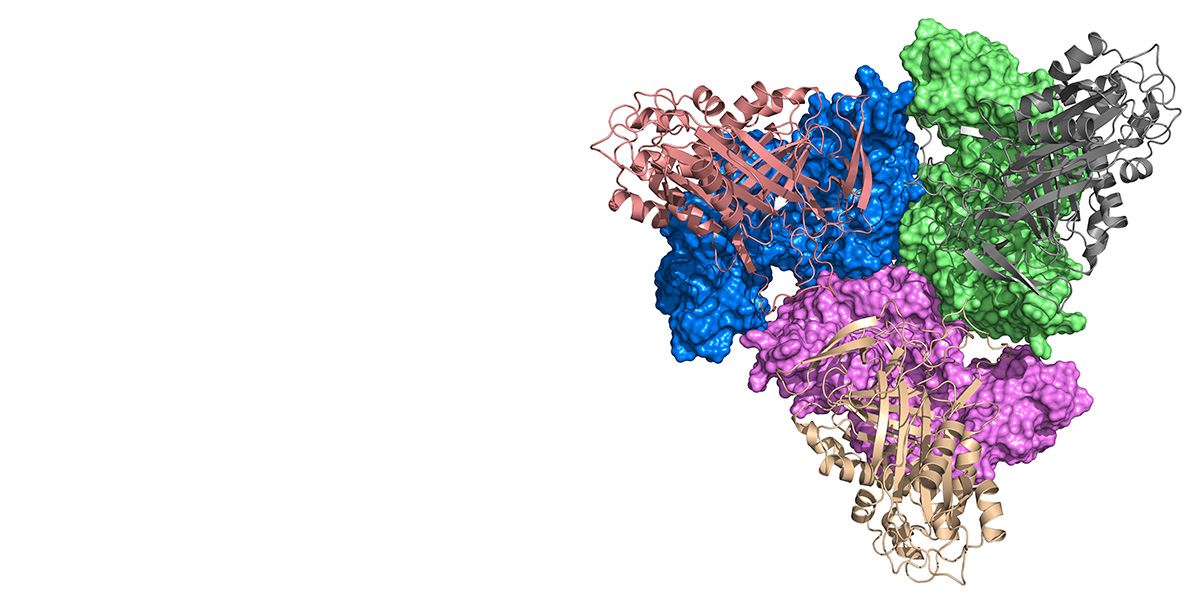
The focus of our research is on the widely conserved HtrA family of serine proteases that are involved in all aspects of ATP-independent protein quality control. We showed that a protein can combine the antagonistic functions of chaperone and protease activities within a single polypeptide.
Furthermore, in collaboration with Tim Clausen (IMP Vienna), it was shown that HtrAs can switch between various oligomeric states and the mechanism of activation by oligomerisation has been elucidated.
In recent years, work on human HTRA1 has revealed its involvement in cancer (as a tumor suppressor), in arthritis (by remodelling of the extracellular matrix) and in Alzheimer's disease (by degrading proteins and protein fragments that aggregate to form senile plaques).
Since a number of years we are using chemical biology approaches, mainly in collaboration with Markus Kaiser (Uni Duisburg-Essen), Biotech and Pharmaceutical companies to generate tools for basic research and for drug development purposes. In addition, the structure of HTRA1 with a bound inhibitor provides leads for the search of HTRA1 modulators that are of therapeutic relevance.
Publications
Beaufort N, Ingendahl L, Merdanovic M, Schmidt A, Podlesainski D, Richter T,
Neumann T, Kuszner M, Vetter IR, Stege P, Burston SG, Filipovic A, Ruiz-Blanco
YB, Bravo-Rodriguez K, Mieres-Perez J, Beuck C, Uebel S, Zobawa M, Schillinger
J, Malik R, Todorov-Völgyi K, Rey J, Roberti A, Hagemeier B, Wefers B, Müller
SA, Wurst W, Sanchez-Garcia E, Zimmermann A, Hu XY, Clausen T, Huber R,
Lichtenthaler SF, Schmuck C, Giese M, Kaiser M, Ehrmann M, Dichgans M. Rational
correction of pathogenic conformational defects in HTRA1.
Nat Commun. 2024 Jul
16;15(1):5944.
Rey J, Breiden M, Lux V, Bluemke A, Steindel M, Ripkens K, Möllers B, Bravo
Rodriguez K, Boisguerin P, Volkmer R, Mieres-Perez J, Clausen T, Sanchez-Garcia
E, Ehrmann M. An allosteric HTRA1-calpain 2 complex with restricted activation
profile.
Proc Natl Acad Sci U S A. 2022 Apr 5;119(14):e2113520119.
Merdanovic M, Burston SG, Schmitz AL, Köcher S, Knapp S, Clausen T, Kaiser M,
Huber R, Ehrmann M. Activation by substoichiometric inhibition.
Proc Natl Acad
Sci U S A. 2020 Jan 6;201918721.
Köcher S, Rey J, Bongard J, Tiaden AN, Meltzer M, Richards PJ, Ehrmann M, Kaiser M. (2017) Tailored Ahp-cyclodepsipeptides as Potent Non-covalent Serine Protease Inhibitors.
Angew Chem Int Ed Engl 56:8555-8558.
Poepsel S, Sprengel A, Sacca B, Kaschani F, Kaiser M, Gatsogiannis C, Raunser S, Clausen T, Ehrmann M. (2015) Determinants of amyloid fibril degradation by the PDZ protease HTRA1.
Nat Chem Biol. 2015 Nov;11(11):862-9.
Mastny M, Heuck A, Kurzbauer R, Heiduk A, Boisguerin P, Volkmer R, Ehrmann M Rodriguez CDA, Rudner DZ, Clausen T. (2013) CtpB assembles a gated protease tunnel regulating cell-cell signaling during spore formation in Bacillus subtilis.
Cell. 155:647–658.
Malet M, Canellas F, Sawa J, Yan J, Thalassinos K, Ehrmann M, Clausen T, and H. Saibil. (2012) Newly folded substrates inside the molecular cage of the HtrA chaperone DegQ.
Nat Struct Mol Biol. 19(2):152-7.
Merdanovic M, Clausen T, Kaiser M, Huber R and M. Ehrmann. (2011) Protein quality control in the bacterial periplasm.
Annu. Rev. Microbiol. 65:149-168.
Kley J, Schmidt B, Boyanov B, Stolt-Bergner PC, Kirk R, Ehrmann M, Knopf RR, Naveh L, Adam Z, Clausen T. (2011) Structural adaptation of the plant protease Deg1 to repair photosystem II during light exposure.
Nat. Struct. Mol. Biol. 18:728-731.
Clausen T, Kaiser M, Huber R and M. Ehrmann. (2011) HtrA proteases: Regulated proteolysis in protein quality control.
Nat. Rev. Mol. Cell Biol.12:152-62.
Trübestein L, Tennstaedt A, Mönig T, Krojer T, Canellas F, Kaiser M, Clausen T, and M. Ehrmann. (2011) Substrate induced remodeling of the active site regulates human HtrA1 activity.
Nat. Struct. Mol. Biol. 18:386-388.
Merdanovic M, Mamant N, Meltzer M, Poepsel S, Auckenthaler A, Melgaard R, Hauske P, Nagel-Steger L, Clarke AR, Kaiser M, Huber R, and M. Ehrmann. (2010) Determinants of structural and functional plasticity of a widely conserved protease chaperone complex.
Nat. Struct. Mol. Biol. 17:837-43.
Krojer T, Sawa J, Schäfer E, Saibil HR, Ehrmann M, and T. Clausen (2008). Structural basis for the regulated protease and chaperone function of DegP. Nature 453, 885-890.
Meltzer M, Hasenbein S, Hauske P, Kucz N, Merdanovic M, Grau S, Beil A, Jones D, Krojer T, Clausen T, Ehrmann M,* and M. Kaiser* (2008). Allosteric activation of HtrA protease DegP by stress signals in bacterial protein quality control.
Angew Chem. Int. Ed. Engl. 47:1332-1334 *corresponding authors.
Hasselblatt H, Kurzbauer R, Wilken C, Krojer T, Sawa J, Kurt J, Kirk R, Hasenbein S, Ehrmann M, and T. Clausen (2007). Regulation of the sE stress response by DegS: How the PDZ domain keeps the protease inactive in the resting state and allows integration of different OMP-derived stress signals upon folding stress.
Genes Dev. 21: 2659-2670.
Hasenbein S, Merdanovic M, and M. Ehrmann (2007). Determinants of regulated proteolysis in signal transduction.
Genes Dev. 21: 6-10.
Ehrmann, M. and T. Clausen (2004). Proteolysis as a regulatory mechanism.
Annu. Rev. Genet. 38: 709-724.
Wilken C, Kitzing K, Kurzbauer R, Ehrmann M, and T. Clausen (2004). Crystal structure of the DegS stress sensor: How a PDZ domain recognizes misfolded protein and activates a protease domain.
Cell 117:483-494, (Highlighted in 2004 Cell 117:417-419).
Clausen T, Southan C, and Ehrmann, M. (2002). The HtrA family of proteases, implications for protein composition and cell fate.
Mol. Cell 10:443-455.
Krojer T, Garrido-Franco M, Huber R, Ehrmann M, and T. Clausen. (2002). Crystal structure of DegP (HtrA) reveals a new protease-chaperone machine.
Nature 416: 455-459, (Highlighted in 2002 Nature Rev. Mol. Cell Biol. 3:310).
Spiess C, Beil A, and M. Ehrmann (1999). A temperature-dependent switch from chaperone to protease in a widely conserved heat shock protein.
Cell 97: 339-347.
Read more




EFRE/JTF Project ClearView: New strategies to fight AMD
The aim of the project is to profile inhibitors developed by the consortium for the protease HTRA1 in order to demonstrate target-mediated activity with patient samples and animal models of age-related macular degeneration and thus pave the way for application.
Download (Flyer)

DFG CRC 1430 Molecular Mechanisms on Cell State Transitions
A6: Implications of deregulated proteostasis in cell state transitions