Group 14 azides
Group 14 azides
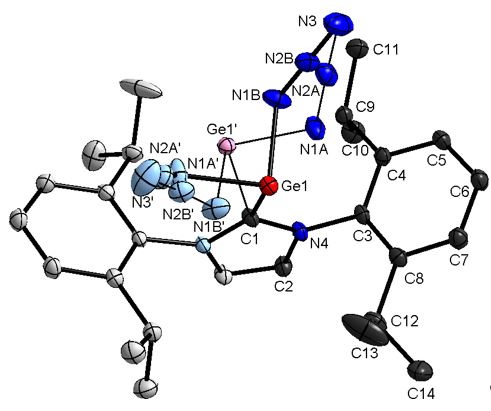
Binary neutral group 14 azides have only been frequently reported. Two homoleptic tetraazides E(N3)4 (E = C, Si) as well as one diazide a-Pb(N3)2 have been synthesized.[1-3] However, a-Pb(N3)2 is the only structurally characterized binary group 14 azides. In addition, the solid state structures of the ionic polyazides [C(N3)3]+, [E(N3)6]2- (E=Si, Ge, Sn, Pb) and [Ge(N3)3]- have been reported.[4] Very recently, Portius reported on the solid state structure of homoleptic salt-like low-valent polyazides E(N3)3]- (E = Ge, Sn),[5] while we reported on the synthesis of binary, base-stabilized group 14 diazides and the structural characterization of an NHC-stabilized germanium diazide IPrGe(N3)2, the first Ge(II) diazide.[6] IPrGe(N3)2 was synthesized by reaction of NaN3 with in situ prepared IPrGeCl2. Its IR spectrum shows a very strong absorption band due to the asymmetric Na-Nb-Ng stretching mode (n = 2075 cm-1) and the Raman spectrum shows two absorption bands due to the asymmetric Na-Nb-Ng stretching mode (n = 2091, 2076 cm-1).
The N-N bond lengths (Na–Nb: 1.206(4), 1.211(4) Å, Nb–Ng: 1.131(4), 1.173(4) Å) of the azido groups are asymmetric and the bond angles (178.4(4), 176.7(4)°) are almost linear as is typical for covalent p-block element azides. The Na-Ge-Na (93.09(12) Å) and the C-Ge-N bond angles (90.42(7), 93.4287)°) are close to 90°, pointing to a large p-orbital contribution of the bonding electron pairs and a high s-character of the electron lone pair. These findings were confirmed by quantum chemical calculations (BP+D3/def2-TZVP), according to wwhich the electron lone pair in this complexes adopts the 4s orbital. The population number of the 4s orbital, which was determined using natural population analysis (NPA), is 1.68.
References
[1] K. Banert, Y.-H. Joo, T. Rüffer, B. Walfort, H. Lang, Angew. Chem. 2007, 119, 1187; Angew. Chem. Int. Ed. 2007, 46, 1168.
[2] a) A. C. Filippou, P. Portius, G. Schnakenburg, M. Davis, K.-D. Wehrstedt, Angew. Chem. Int. Ed. 2010, 49, 43; b) E. Wiberg, H. Michaud, Z. Naturforsch. B 1954, 9, 500; c) R. Herges, F. Starck, J. Am. Chem. Soc. 1996, 118, 12752; e) G. Maier, H. P. Reisenauer, J. Glatthaar, Organometallics 2000, 19, 4775.
[3] a) L. V. Azaroff, Z. Kristallogr. Kristallgeom. Kristallphys. Kristchem. 1956, 107, 362; b) C. S. Choi, E. Prince, W. L. Garrett, Acta Crystallogr. Sect. B 1977, 33, 3536.
[4] a) A. C. Filippou, P. Portius, D. U. Neumann, K.-D. Wehrstedt, Angew. Chem. Int. Ed. 2000, 39, 4333; b) U. Müller, H. Bärnighausen, Acta Crystallogr. Sect. B 1970, 26, 1671; c) M. A. Petrie, J. A. Sheehy, J. A. Boatz, G. Rasul, G. K. S. Prakash, G. A. Olah, K. O. Christe, J. Am. Chem. Soc. 1997, 119, 8802; d) A. C. Filippou, P. Portius, G. Schnakenburg, J. Am. Chem. Soc. 2002, 124, 12396; e) D. Fenske, H.-D. Dörner, K. Dehnicke, Z. Naturforsch. B 1983, 38, 1301; f) K. Polborn, E. Leidl, W. Beck, Z. Naturforsch. B 1988, 43, 1206.
[5] B. Peerless, T. Keane, A. J. H. M. Meijer, P. Portius, Chem.Commun. 2015, 51, 7435.
[6] B. Lyhs, D. Bläser, C. Wölper, S. Schulz, R. Haack, G. Jansen, Inorg. Chem. 2013, 52, 7236.
For more: Group 15 azides
Go back: Covalent Main group element azides