Synthesis and Reactivity of low-valent group 15 compounds
Introduction
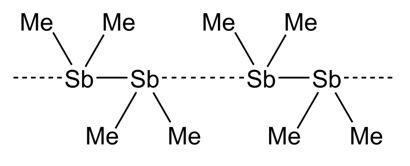
Tetraalkyldistibines R4Sb2 such as Me4Sb2 and Et4Sb2 are long known low-valent organoantimony compounds. Me4Sb2 for instance was synthesized for the first time almost eighty years ago by reaction of an Sb-mirror with methyl radicals.[1,2] Since then, alternative synthetic pathways were established and their structures were investigated, in detail.[3]
Distibines Sb2R4 and dibismuthines R4Bi2 tend to form intermolecular interactions in the solid state, yielding chain-like structures with E···E bond lengths below the sum of the van-der-Waals radii (4.12 Å).[4]. The formation of intermolecular interactions is typically observed for compounds containing small organic substituents, whereas sterically demanding substituents hamper the formation of intermolecular contacts.
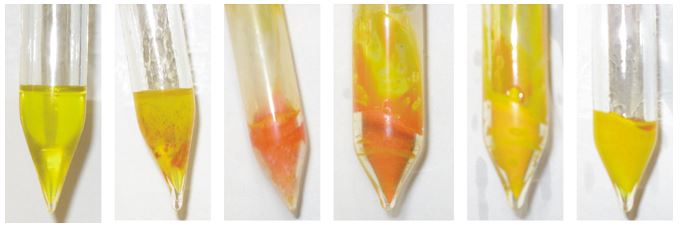
These interactions are rather weak and are often interrupted upon melting, resulting in a bathochromic shift between fluid and solid phases, the so-called ''thermochromic'' behavior.[5] For instance, Me4Sb2 is yellow in the solid state in liquid nitrogen, red close to its melting point of 17 °C, again yellow in the fluid state but colourless in the gas phase.[6] We recently studied the solid state structures of Et4Sb2 and Et4Bi2.[7]
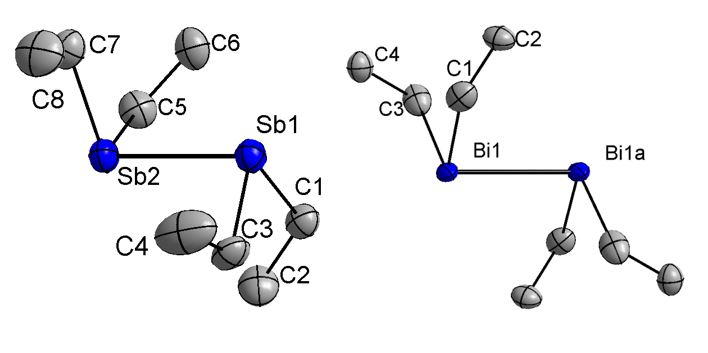
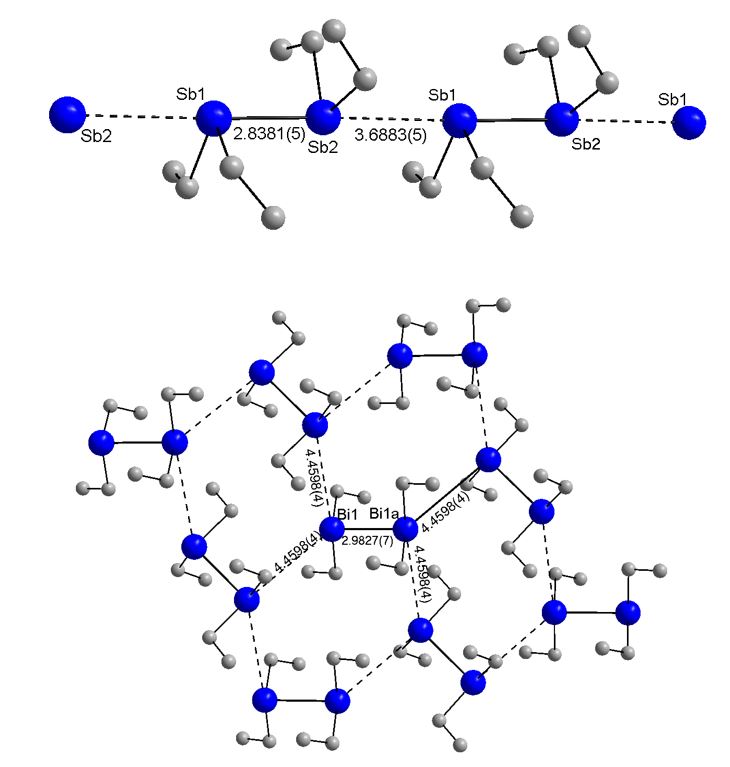
The crystals were obtained by in situ crystallization on the diffractometer using an IR laser-asssited technique. Et4Sb2 forms endless chains via short intermolecular Sb···Sb contacts of 3.688(1) Å between two distibine units, which agrees very well with values of reported for Me4Sb2 (3.645, 3.678 Å) and [(HC=CMe)2]2Sb2 (3.63 Å). In contrast, Et4Bi2 doesn't form isolated Bi chains but exists as rather isolated molecules and the closest intermolecular Bi···Bi distance of 4.4598(4) Å is longer than the sum of the van-der-Waals radii (4.14 Å).
According to these structural findings, it isn't surprising that Et4Sb2 shows thermochromic behavior, while Et4Bi2 only shows an intensification of the red color to a deep red color upon cooling to -103° C.
Refences
[1] For a historical review see: D. Seyferth, Organometallics 2001, 20, 1488.
[2] a) F. A. Paneth, Trans. Faraday Soc. 1934, 30, 179; b) F. A. Paneth, H. Loleit, J. Chem. Soc. 1935, 366.
[3] For review articles see: a) H. J. Breunig, R. Rössler, Coord. Chem. Rev. 1997, 163, 33; b) C. Silvestru, H. J. Breunig, H. Althaus, Chem. Rev. 1999, 99, 3277; c) H. J. Breunig, R. Rössler, Chem. Soc. Rev. 2000, 29, 403; d) Y. Matano, T. Ikegami, in Organobismuth Chemistry, ed. H. Suzuki, Y. Matano, Elsevier, Amsterdam, 2001, p. 107ff; e) G. Balázs, H. J. Breunig, in Unusual Structures and Physical properties in organometallic Chemistry, ed. M. Gielen, R. Willem, B. Wrackmeyer, J. Wiley & Sons, West Sussex (GB), 2002, p. 387ff; f) L. Balázs, H. J. Breunig, Coord. Chem. Rev. 2004, 248, 603; g) H. J. Breunig, Z. Anorg. Allg. Chem. 2005, 631, 621.
[4] Mantina, M.; Chamberlin, A. C.; Valero, R.; Cramer, C. J.; Truhlar, D. G. J. Phys. Chem. A 2009, 113, 5806.
[5] a) Ashe, III, A. J.; Ludwig Jr., E. G.; Oleksyszyn, J.; Huffman J. C. Organometallics 1984, 3, 337; b) Mundt, O.; Riffel, H.; Becker, G.; Simon, A. Z. Naturforsch. 1984, 39b, 317; c) Mundt, 0.; Riffel, H.; Becker, G.; Simon, A. Z. Naturforsch. 1988, 43b, 952; d) Breunig, H. J. Z. Anorg. Allg. Chem. 2005, 631, 621; e) Breunig, H. J.; Lork, E.; Moldovan, O.; Rat, C. I. J. Organomet. Chem. 2008, 693, 2527; f) Monakhov, K. Y.; Zessin, T.; Linti G., Eur. J. Inorg. Chem. 2010, 322.
[6] A. J. Ashe, III, E. G. Ludwig, Jr., J. Oleksyszyn, J. C. Huffman Organometallics 1984, 3, 337.
[7] a) Kuczkowski, A.; Heimann, S.; Weber, A.; Schulz, S.; Bläser, D.; Wölper, C. Organometallics 2011, 30, 4730; b) Schulz, S.; Heimann, S.; Kuczkowski, A.; Wölper, C.; Bläser, D. Organometallics 2013, 32, 3391.