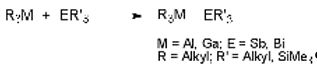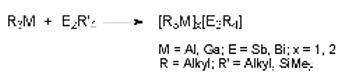Synthesis of Group 13-15 compounds
Synthesis of Group 13-15 Compounds
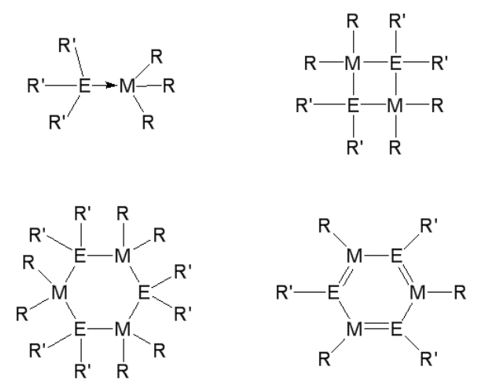
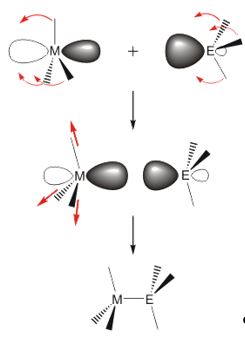
Complexes containing elements of the group 13 and 15, so-called III/V compounds, are studied for nearly 200 years. The first compound of this type, F3B-NH3, which was already synthesized in 1809 by Gay-Lussac, is the classical representative of a Lewis acid-base adduct. In 1926 Stock and Pohland synthesized borazine, which is also called "inorganic benzene". Since that time, numerous Lewis acid-base adducts R3M←ER'4 and heterocycles of the type [R2MER'2]x and [RMER ']3 have been synthesized and structurally characterized.
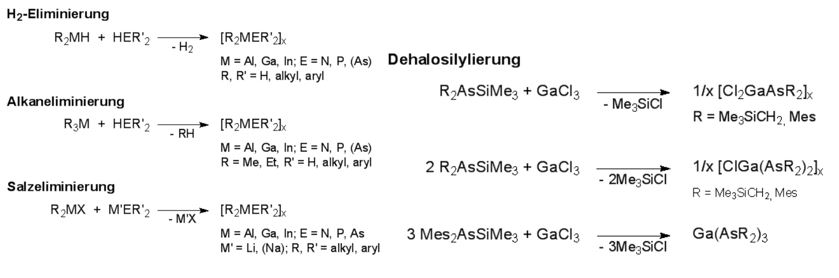
The adducts are easily accessible by reaction of the Lewis acid R3M with the Lewis base ER'3, while several general pathways for the synthesis of heterocycles have been established. Compounds with light elements of the group 15 (N, P, As) are typically synthesized by alkane and salt elimination reactions as well as by dehalosilylation reaction.
In contrast, analogous Lewis acid-base adducts and heterocycles with the heaviest elements of the group 15, Sb and Bi, have been unknown for a long time. This is based on the one hand on the relatively low Lewis-basicity of organostibines and -bismuthines ER3 (E = Sb, Bi) and, on the other hand, the failure of established synthetic methods, which can`t be transferred to the heterocycles synthesis of the heavy group 15 homologues.
We particularly concentrated on the synthesis, structure and reactivity of Lewis acid-base adducts of the type R3M-ER'3 and [R3M]2[E2R'4] containing the heaviest group 15 elements (M = Al, Ga, In, E = Sb, Bi). We are established new pathways for the synthesis of ME-heterocycles including the heaviest elements of the group 13 (In, Tl) and 15 elements (Sb, Bi) and examine their structures (solution or solid state) and reactivity.
Group 13/15 Lewis acid-base adducts
Lewis acidic group 13 compounds of the type R3M (M = B, Al, Ga, In; R = H, alkyl, halogen) react with Lewis bases with formation of Lewis acid-base adducts. This fundamental reaction has been studied intensively for a long time and large numbers of compounds have been synthesized and characterized by different spectroscopic techniques (NMR, IR, Raman) and diffraction methods (XRD, ED), while quantum chemical calculations provided a profound knowledge of their bonding situation.
The main focus of these studies was on amine and phosphine adducts. Recently published research results on the use of the amine-borane adduct H3BNH3 as hydrogen storage material as well as the hydrogen activation of so-called "frustrated Lewis pairs FLP" has given another push to this field of research.[1,2] In contrast, adducts with triorganostibines and bismuthines have been studied to a far lesser extent.[3] This is mainly due to their lower stability as a result of the relatively low Lewis basicity (decreasing σ-donor ability). In particular bismuthines BiR3 are very weak σ-donors as a result of the lanthanide contraction as well as relativistic effects that increase the contraction of the s-orbital and its energy reduction (stability). The donor ability of triorganopenteles ER3 may be substantially controlled by the substitution pattern. The p-character of the electron lone pair of trialkylpenteles ER3 increases with increasing steric demand of the alkyl ligands, hence leading to an enhanced Lewis basicity. However, this increase of Lewis basicity (donor ability) faces repulsive interactions with the acceptor molecule, so that the resulting adducts are not necessarily thermodynamically stable. In particular, adducts with small central atoms are extremely sensitive to repulsive steric interactions.
We synthesize group 13/15-adducts of the heaviest homologues of the group 15 elements by using sterically demanding-substituted triorganostibines and bismuthines and investigated their bonding situation in the solid state by X-ray diffraction and their stability in solution by temperature-dependent NMR spectroscopy. The preparative studies are accompanied by theoretical investigations concerning their structure and stability.
Adducts of triorganopenteles ER3
Triorganostibines and -bismuthines ER3 react with trialkylalanes and -gallanes under strict exclusion of air and moisture to the corresponding Lewis acid-base adducts.[4]
Many of these air-sensitive and temperature-labile compounds have been characterized by single-crystal X-ray diffraction. The central M-E bond length typically ranges from 280 to 300 pm (M-Sb) or 290 to 315 pm (M-Bi), and therefore clearly deviate from the sum of the covalent radii of 265 (M-Sb) or 274 pm (MBi).
The strength of the acid-base interactions can be estimated from structural parameters using a model that was initially introduced by Haaland and later on verified by other research groups.[5] The adduct formation is accompanied by an increase of the M-C bond length and a decrease of the C-M-C bond angle and the relative strength of the Lewis acid-base interaction is reflected by the extent of the change.
The thermodynamic stability of these adducts in solution was investigated by temperature-dependent NMR spectroscopy and the dissociation energies of the stibine adducts were found to range from 5 to 10 kcal/mol, while the bismuthine adducts are thermodynamically less stable. These values were verified by quantum chemical calculations.[6]
Adducts of distibines and dibismuthines E2R4
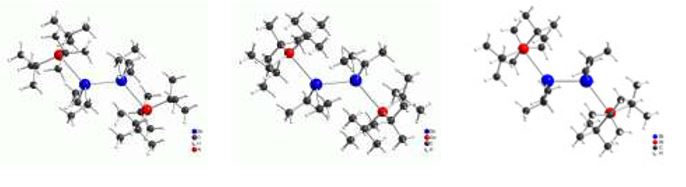
Distibines and dibismuthines R'2E–ER'2, in which the central metal atoms adopt the formal oxidation state +2, have two Lewis-basic centers. Despite their relatively weak E-E bond, several stable bis-adducts [R3M]2[E2R'4] (E = Sb, Bi) as well as the mono-adduct [t-Bu3Al][Sb2(i-Pr)4] were obtained from reactions with trialkylalanes and -gallanes MR3.[7,8]
The trialkylalanes and-gallanes in bis-adducts adopt a trans-orientation position as is expected. The central E-E bond is almost unchanged compared to the starting distibines and dibismuthines. The compounds are only temporarily stable in solution and tend to form MSb heterocycles.
References
[1] (a) F. H. Stephens, R. T. Baker, M. H. Matus, D. J. Grant, D. A. Dixon, Angew. Chem. Int. Ed. 2007, 46, 746. (b) F. H. Stephens, V. Pons, R. T. Baker, J. Chem. Soc. Dalton Trans. 2007, 2613.
[2] (a) G. C. Welch, R. R. San Juan, J. D. Masuda, D. W. Stephan, Science 2006, 314, 1124. (b) V. Sumerin, F. Schulz, M. Nieger, M. Leskelä, T. Repo, B. Rieger, Angew. Chem. 2008, 120, 6090.
[3] W. Levason, G. Reid, Coord. Chem. Rev. 2006, 250, 2565.
[4] (a) S. Schulz, M. Nieger, Organometallics 1999, 18, 315. (b) S. Schulz, M. Nieger, J. Chem. Soc. Dalton Trans. 2000, 639. (c) S. Schulz, A. Kuczkowski, M. Nieger, J. Organomet. Chem. 2000, 604, 202. (d) S. Schulz, F. Thomas, A. Kuczkowski, M. Nieger, Organometallics 2000, 19, 5758.
[5] (a) A. Haaland, Angew. Chem., Int. Ed. Engl. 1989, 28, 992. (b) A. Haaland in Coordination Chemistry of Aluminum, Ed. G.H. Robinson, VCH Verlagsgesellschaft, Weinheim 1993.
[6] A. Kuczkowski, S. Schulz, M. Nieger, P. R. Schreiner, Organometallics 2002, 21, 1408.
[7] (a) A. Kuczkowski, S. Schulz, M. Nieger, Angew. Chem. 2001, 113, 4351. (b) A. Kuczkowski, S. Schulz, M. Nieger, Organometallics 2001, 20, 2000. (c) D. Schuchmann, A. Kuczkowski, S. Fahrenholz, S. Schulz, U. Flörke, Eur. J. Inorg. Chem. 2007, 931.
[8] A. Kuczkowski, S. Fahrenholz, S. Schulz, M. Nieger,