Reactivity of group 15 complexes
Reactivity of group 15 complexes
Trialkylstibines and bismuthines R3E (E = Sb, Bi) were intensely studied in reactions with Lewis acidic group 13 trialkyls, yielding Lewis acid base adducts. Adduct formation was also observed in reactions of distibines and dibismuthines with group 13 metal complexes. Recently, we studied oxidation reactions of R3E as well as insertion reactions of low valent distibines and dibismuthines R4E2 with group 16 elements as well as organometallic group 16 element compounds.
Oxidation reactions of R3Sb
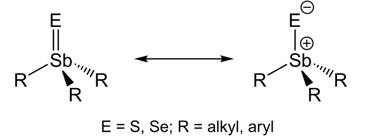
Even though chalcogenostiboranes R3SbE (E = O, S, Se; R = alkyl, aryl) have been initially prepared more than 150 years ago by Carl Jakob Löwig and Eduard Schweizer by redox reaction of Et3Sb with elemental sulfur and selenium,[1] reports on solid state structures of R3SbE bearing an unsupported terminal Sb=E double bond are almost unknown, Ph3SbS represents the only structurally characterized triorganylthiostiborane.[2,3] Pebler et al. described the short Sb-S bond in Ph3SbS (2.244(1) Å) as partial double bond, resulting from a dπ–pπ interaction,[3] while Otera et al. investigated the bonding situation in Me3SbS and calculated a positive charge of +0.6 at the Sb atom, indicating the Sb-S bond to be best described as polar single bond with some ionic stabilization.[4] Vibrational spectroscopy was used to clarify the bonding situation. The Sb-E stretching vibration frequencies for a Sb-E single bond and Sb=E double bond were calculated using Gordy's rules and compared with the experimental values for Me3Sb=S, Et3Sb=S and Et3Sb=Se. However, since the experimental values fall in between the calculated values, a clear distinction between both bonding situations was not possible. Therefore, it is still unclear whether the Sb-E bond in chalcogenostiboranes should be described as a polar single bond or as a real double bond (Scheme 1).
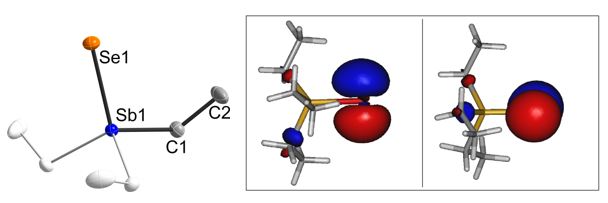
We synthesized Et3Sb=S and Et3Sb=Se, determined their solid state structures by single crystal X-ray diffraction and investigated the bonding situation by quantum chemical calculations.[5] The Sb-S bond length of Et3Sb=S (2.381(7) Å) is in between the calculated values for the Sb-S single bond (Sb–S 2.43 Å) and the Sb=S double-bond (2.27 Å) and significantly elongated compared to that in Ph3Sb=S (2.244(1) Å). Et3Sb=Se is the first structurally characterized trialkylselenostiborane and shows the shortest Sb–Se bond length (2.4062(8) Å) reported, to date, which perfectly agrees with the calculated Sb=Se double bond (Sb–Se 2.40 Å). However, according to quantum chemical calculations, the shortening of the Sb-E bonds in Et3Sb=S and Et3Sb=Se results from a strong electrostatic interaction between the central Sb atom and the chalcogen bonding partner as both bonds are strongly polarized, Sb(δ+)–E(δ−) (vide infra). Natural bond orbital (NBO) analysis suggests the presence of three lone pair orbitals on the chalcogen atom. One of the lone pair orbitals consists of the valence shell s orbital (S: 89%; Se: 92%), the remaining two are pure valence shell p orbitals orthogonal to the Sb-E bond, thus precluding the existence of a Sb-E double bond in both cases due to p-interactions.
References
[1] C. Löwig, E. Schweizer, Liebigs Ann. Chem. 1850, 75, 315.
[2] a) A. Hantzsch, H. Hibbert, Ber. Dtsch. Chem. Ges. 1907, 40, 1508; b) W. J. C. Dyke, W. J. Jones, J. Chem. Soc. 1930, 1921; c) R. A. Zingaro, A. Merijanian, J. Organomet. Chem., 1964, 1, 369; d) G. N. Chremos, R. A. Zingaro, J. Organomet. Chem. 1970, 22, 637.
[3] L. Kaufmann, Chem. Ber. 1908, 41, 2762; W. J. Lile, R. C. Menzies, J. Chem. Soc. 1950, 617.
[4] a) J. Pebler, F. Weller, K. Dehnicke, Z. Anorg. Allg. Chem. 1982, 492, 139; b) F. Weller, K. Dehnicke, Naturwissenschaften 1981, 68, 523; c) G. S. Zhdanov, V. A. Pospelov, M. M. Umanski, V. P.Glushkova, Dokl. Akad. Nauk SSSR (Russ.) (Proc. Nat. Acad. Sci. USSR) 1953, 92, 983.
[5] S. Heimann, D. Bläser, C. Wölper, R. Haack, G. Jansen, S. Schulz, Dalton Trans, 2014, 43, 14772.