Reactivity of Organozinc Complexes
Reactivity of organozinc complexes
Synthesis of N,N'-chelating complexes
Organometallic complexes of s-, p-, d-and f-block metals with anionic N, N'-chelate ligands, such as b-diketiminate, guanidinate- and amidinate ligands have been studied for a long time.[1-4]. Their steric and electronic properties can be tuned by variation of the substitution pattern. They show a remarkable bond variability and can act as bidentate (η2) and bridging monodentate (μ-η1-η1) four-electron donors. b-diketiminate zinc complexes LZnR (L = β-diketiminate) are intensely studied due to their promising potential as highly active catalysts in the ring-opening polymerization (ROP) of cyclic esters,[5] particularly of lactides,[6] as well as in the copolymerization of CO2 with epoxides.[7]
Synthesis and structure of amidinate- and β-diketiminate zinc complexes
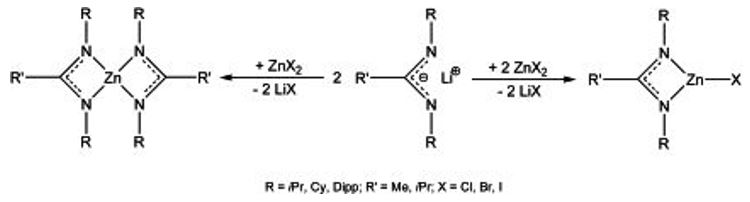
Hetero- and homoleptic amidinate zinc complexes of the general type LZnX and L2Zn (L = amidinate, ß-diketiminate; X = alkyl, aryl, halide, amide) are synthesized by salt elimination reactions.[8,9]
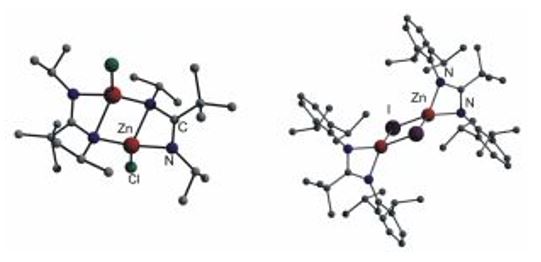
Monoamidinate zinc complexes typically form centrosymmetric dimers with bridging nitrogen atoms in which the amidinate ligand coordinates as a chelating (η2) or bridging (μ-η1-η1) unit. [t-BuC(NDipp)2Zn(μ-I)]2, containing the sterically demanding Dipp substituent (Dipp = 2,6-i-Pr2C6H3), forms a iodine-bridged dimeric complex.[8,9,10]
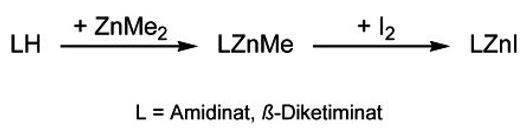
We established a straightforward pathway for the synthesis of heteroleptic amidinate and b-diketiminate zinc complexes based on the reaction of ZnMe2 and LH.[10,11]
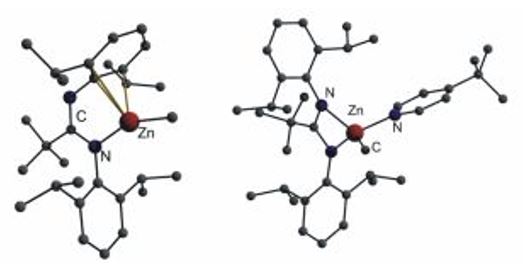
[t-BuC(NDipp)2]ZnMe coordinates in a rather unexpected η1-N,η3-arene fashion. While this coordination mode has been previously observed for large and, according to the HSAB concept, rather soft metals such as K, In and Tl,[12,13] it is rather unexpected for Zn(II) centers. Addition of the Lewis base t-butyl pyridine leads to a structural rearrangement to the common N,N'-chelating binding mode.[10]
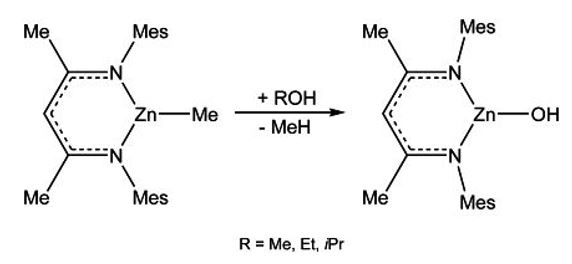
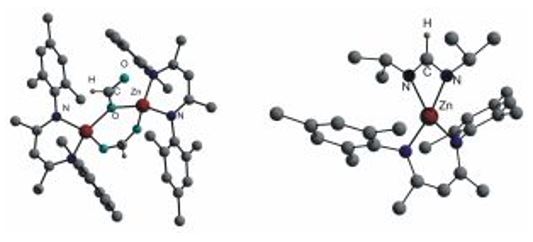
The heteroleptic complexes LZnX are suitable for substitution reactions. MesnacnacZnMe (Mesnacnac = [HC{C(Me)N(2,4,6-Me3C6H2)}2]) reacts with alcohols ROH (R = Me, Et, i-Pr) with elimination of methane and subsequent formation of the corresponding zinc alkoxides, which typically adopt dimeric structures with bridging OR-groups in the solid state.[14,15]
MesnacnacZnCl reacts with NaH or KN(i-Pr)HBH3 with formation of the hydride complex MesnacnacZnH. Insertion reactions of heterocumulenes such as CO2, C(Ni-Pr)2 and t-BuNCO into the Zn–H bond yields the corresponding formato, formamido and formamidinato complexes.[16-18] The formato complex forms an unsymmetrically bridged dimer exhibiting a syn-syn-(μ-η2) and a bridging (μ-η1) HCO2 unit, whereas MesnacnacZnCH(i-PrN)2 with the sterically demanding i-Pr groups is a monomer with a chelating η2-(Ni-Pr)2CH. Comparable insertion reactions of heterocumulenes into Zn-carbon bonds were observed.[19-21]
References
[1] L. Bourget-Merle, M. F. Lappert, J. R. Severn, Chem. Rev. 2002, 102, 3031.
[2] P. J. Bailey, S. Pace, Coord. Chem. Rev. 2001, 214, 91.
[3] a) J. Barker, M. Kilner, Coord. Chem. Rev. 1994, 133, 219; b) F. T. Edelmann, Coord. Chem. Rev. 1994, 137, 403.
[4] M. P. Coles, J. Chem. Soc., Dalton Trans. 2006, 985.
[5] a) J. Prust, H. Hohmeister, A. Stasch, H. W. Roesky, J. Magull, E. Alexopoulos, I. Usón, H.-G. Schmidt, M. Noltemeyer, Eur. J. Inorg. Chem. 2002, 2157; b) S. Aboulkacem, W. Tyrra, I. Pantenburg, Z. Anorg. Allg. Chem. 2003, 629, 1569.
[6] a) M. Cheng, A. B. Attygalle, E. B. Lobkovsky, G. W. Coates, J. Am. Chem. Soc. 1999, 121, 11583; b) S. D. Allen, D. R. Moore, E. B. Lobkovsky, G. W. Coates, J. Am. Chem. Soc. 2002, 124, 14284; c) M. H. Chisholm, J. C. Gallucci, K. Phomphrai, Inorg. Chem. 2005, 44, 8004.
[7] a) B. Y. Liu, C. Y. Tian, L. Zhanq, W. D. Yan, W. J. Zhanq, J. Polym. Sci. Part A: Polym. Chem. 2006, 44, 6243; b) M. Kröger, C. Folli, O. Walter, M. Döring, Adv. Synth. Cat. 2006, 348, 1908.
[8] T. Eisenmann, J. Khanderi, S. Schulz, U. Flörke, Z. Anorg. Allg. Chem. 2008, 634, 507.
[9] S. Schmidt, S. Schulz, M. Bolte, Z. Anorg. Allg. Chem. 2009, 635, 2210.
[10] S. Schmidt, S. Schulz, D. Bläser, R. Boese, M. Bolte, Organometallics, 2010, 29, 6097.
[11] S. Schulz, T. Eisenmann, U. Westphal, S. Schmidt, U. Flörke, Z. Anorg. Allg. Chem. 2009, 635, 216.
[12] J. Baldemus, C. Berghof, M. L. Cole, D. J. Evans, E. Hey-Hawkins, P. C. Junk, Dalton Trans. 2002, 2802.
[13] C. Jones, P. C. Junk, J. A. Platts, D. Rathmann, A. Stasch, Dalton Trans. 2005, 2497.
[14] S. Schulz, T. Eisenmann, D. Bläser, R. Boese, Z. Anorg. Allg. Chem. 2009, 635, 995.
[15] S. Schulz, T. Eisenmann, D. Schuchmann, M. Bolte, M. Kirchner, R. Boese, J. Spielmann, S. Harder, Z. Naturforsch. 2009, 64b, 1397.
[16] S. Schulz, T. Eisenmann, S. Schmidt, D. Bläser, U. Westphal, R. Boese, Chem. Commun. 2010, 46, 7226.
[17] a) T. Eisenmann, J. Khanderi, S. Schulz, U. Flörke, Z. Anorg. Allg. Chem. 2008, 634, 507. b) S. Schulz, M. Münch, U. Flörke, Z. Anorg. Allg. Chem. 2008, 634, 2221.
[18] G. Bendt, S. Schulz, J. Spielmann, S. Schmidt, D. Bläser, C. Wölper, Eur. J. Inorg. Chem. 2012, 23, 3725.
[19] S. Schmidt, B. Gutschank, S. Schulz, D. Bläser, R. Boese, C. Wölper, Eur. J. Inorg. Chem. 2011, 28, 4464.
[20] S. Schmidt, R. Schäper, S. Schulz, D. Bläser, C. Wölper, Organometallics 2011, 30,1073.
[21] S. Schulz, S. Schmidt, D. Bläser, C. Wölper, Z. Anorg. Allg. Chem. 2012, 638, 1705.