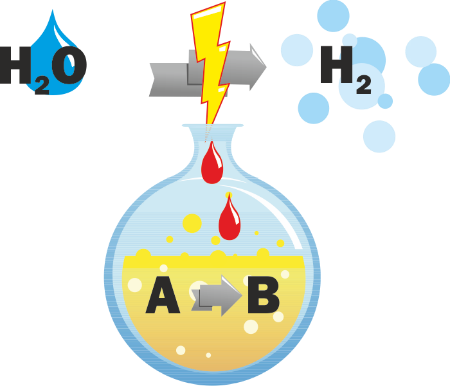
A future sustainable energy system based on hydrogen is inevitable and the generation of this energy carrier will be definitely achieved by electrolysis. The oxygen evolution in the course of water electrolysis represents still a challenge which consumes a significant amount of electric power due to high overpotentials. Alternative anodic conversions which do not liberate dioxygen but serve as useful and significant anodic transformations represent an innovative solution. For this aim, two different approaches are pursued: The development and establishing of electrochemical oxidation reactions with a high impact and technical relevance. Among others, the anodic functionalization of methane, the oxidation of alcohols, specifically glycerol under formation of e.g. lactic acid, the oxidation of hydroxymethylfurfural to the renewable platform chemical 2,5-furan dicarboxylic acid, as well as the oxidation of amines to amine-N-oxides represent such challenging oxidative conversions. An alternative approach will anodically generate oxidizing equivalents which can later on be exploited to a variety of chemical applications. This strategy avoids the selectivity issues of rather complex molecules at the anode. In addition, it will establish a general route which opens up multipurpose applications and compensate fluctuations in the electric current consumptions since these oxidizers can be stored. In order to tackle these challenges in a knowledge-driven way, important tools of investigation such as operando electrochemistry/spectroscopy and the influence of the electrode morphology will be addressed. This research unit will bridge the gap from fundamentals of organic electrochemistry and electrocatalysis to prep-type electrolysis including initial steps to upscaling.