CRC 1093 - About
Collaborative Research Centre 1093:
Supramolecular Chemistry on Proteins
Protein function is intimately connected with supramolecular chemistry: Substrate processing and various levels of regulation involve specific noncovalent interactions. Likewise, protein folding, assembly and protein-protein interactions are governed by intermolecular forces and their specific inhibition or support represents a very promising area for external interference, with profound mechanistic and therapeutic implications.
The CRC aims at a deeper understanding of the underlying principles of protein recognition and functional modulation by supramolecular ligands. Combining different expertise from synthetic and supramolecular chemistry, molecular and cell biology with computational chemistry, bioinformatics and structural biology provides a highly interdisciplinary research environment for this endeavor. Chemists develop novel concepts for protein recognition and modulation by supramolecular ligands which biologists apply to answer biological questions, which can best be tackled with supramolecular chemistry.
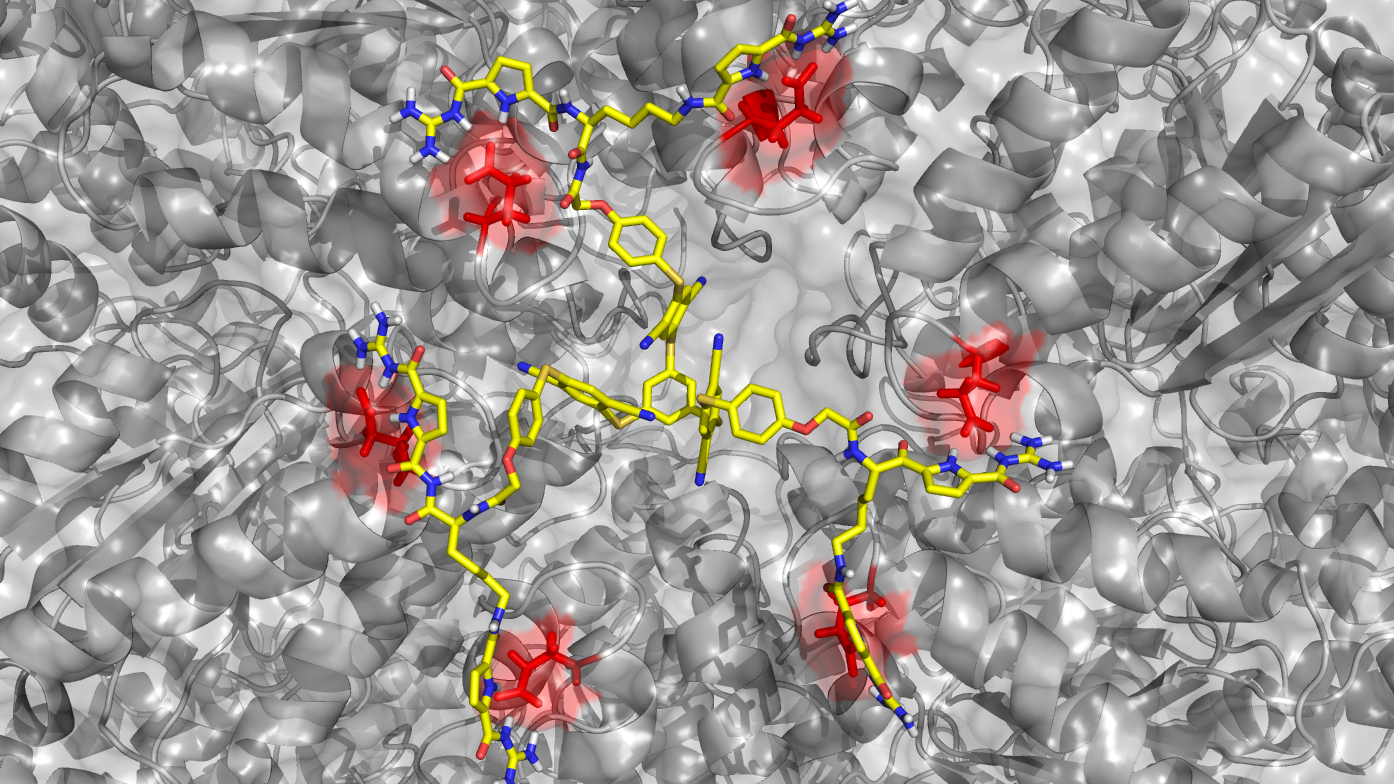
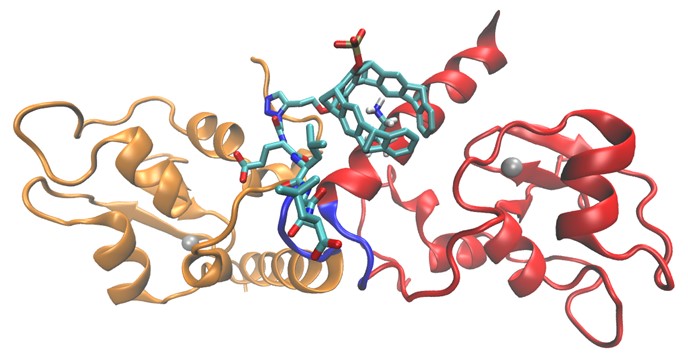
To address e.g. protein-protein interactions the supramolecular ligands need to bind in shallow grooves or large pores on the protein surface which is often more challenging than the development of traditional enzyme inhibitors which bind in deep pockets at active sites of a protein. We use tailor-made recognition motifs for specific protein epitopes, combinatorial approaches or dendrimers, affinity copolymers, precision macromolecules or ultrasmall nanoparticles as scaffolds for binding epitopes on proteins (area A). Ligands are synthesized and first characterized for their binding profile using isolated proteins and then passed on to the biologists where they are employed to study inter alia biochemical mechanisms of allosteric and cooperative protein activation, the function of protein pores or the importance of protein-protein interactions e.g. in chaperones, proteases, transport systems and mitotic regulators (area B). Two structural biology projects, X-ray crystallography and NMR spectroscopy (area Z) give structural information on the ligand protein complexes to deepen the molecular understanding of the observed biological effects and to allow further optimization of the ligands. Experimental structural analyses are further complemented and extended by theoretical calculations on essential aspects of protein surface recognition by the supramolecular ligands.
more about „Supramolecular Chemistry on Proteins”