Material Synthesis
1. Gas Phase Processes
1.1 MOCVD Process
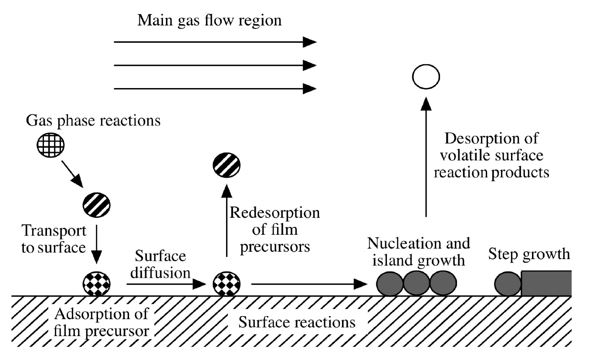
III/V materials are typically mainly produced by Liquid Phase Epitaxy (LPE), Molecular Beam Epitaxy (MBE) and Chemical Vapor Deposition (CVD). The CVD process, which was first described by Manasevit,[1] is a very complex process and the deposition of high quality material films requires the full understanding and control of various chemical and physical processes both in the gas phase and on the surface of the substrate.
1.2 ALD Process
Atomic layer deposition is a versatile technique for the deposition of thin films. The ALD-process was first described in 1974 by Suntola and Antson for the deposition of a thin ZnS film using H2S and ZnCl2 as precursors.[2] While this technique was initially used for the deposition of electroluminescent films on large surfaces,[3] it is nowadays applied for the deposition of metaloxides, -nitrides, -sulfides, metals, polymers and inorganic-organic hybridmaterials, which find widespread technical applications in microelectronics, medicine and biology.[4] One of the big advantages of the ALD-technique consists of the ability to coat 3D structures homogenously.
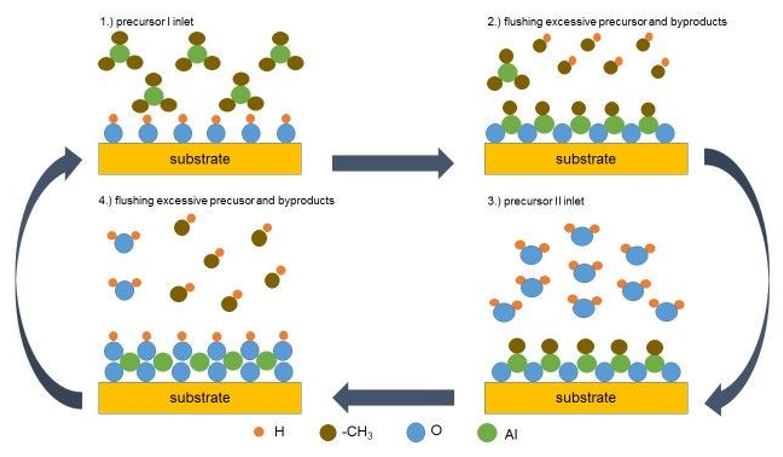
The ALD deposition is a self-limiting process containing reactions between precursor molecules from the gas phase and the substrate. A typical ALD-cycle is illustrated in figure 1, showing the formation of Al2O3 by sequential reaction of Me3Al and H2O.[5]
Precursor I (Me3Al) is induced into the reaction chamber and reacts with the functionalized substrate's surface (typically SiO2 with OH-groups) with subsequent elimination of CH4 (step 2). The excess of the precursor is then pumped of, followed by indroduction of precursor II (H2O) into the chamber, which immediately reacts with the Al-Me functionalized surface via elimination of methane (step 4). Another purging step is then carried out in order to avoid undesirable reactions of the byproducts and with the surface as well. In average the growth rate amounts to 0.01 – 2 Å per cycle, depending on the precursors.
2. Wet Chemical Synthesis
Widely applied wet-chemical routes for the synthesis of nanoparticles are the polyol process and the DMF-mediated reduction, which either use diethyleneglycol (DEG) or dimethylformamide (DMF) are reducing solvents, as well as the by the seeded-growth method.[6] The thermolytic reduction of the inorganic or organometallic precursor typically requires high temperatures, which are either achieved using a heating mantle or an oil bath or through the so-called "hot-injection" method, in which a solution of the precursor is rapidly injected into a hot solvent. Instead of a pure organic solvent one can also use a solvent mixture with octadecylamine (ODA), which can serve as solvent, surfactant, and reducing agent, and often allows the controlled preparation of a wide range of metals, metal oxide, and semiconducting nanoparticles.[7]
While inorganic precursors have a long standing tradition in material synthesis, in recent years many researchers started to focus on the use of organometallic precursors since these are typically thermodynamically less stable and allow material synthesis at low temperatures.[8] In addition, alternate solvents such as ionic liquids (IL) are nowadays widely applied for the size- and form-selective synthesis of a wide varity of materials.[9]
References
[1]H. M. Manasevit, Appl. Phys. Lett . 1968, 12, 156.
[2] T. Suntola, J. Hyvärinen, Ann. Rev. Mater. Sci. 1985, 15, 177.
[3] T. Suntola in "Handbook of crystal growth", (ed. D. T. J. Huerle), Vol. 3, Elsevier, Amsterdam, 1994.
[4] S. A. Skoog, J. W. Elam, R. J. Narayan, Int. Mat. Rev. 2013, 58, 113.
[5] N. Pinna, M. Knez (Eds.) in Atomic Layer Deposition of Nanostructured Materials, Wiley, Weinheim, 2002.
[6] I. Pastoriza-Santos, L. M. Liz-Marzán, Adv. Funct. Mater. 2009, 19, 679.
[7] a) D. Wang, Y. Li, Inorg. Chem. 2011, 50, 5196; b) M. H. Huang, P.-H. Lin, Adv. Funct. Mater. 2012, 22, 14; c) H. Zhang, M. Jin, Y. Xia, Angew. Chem. Int. Ed. 2012, 51, 7656; Y. Xia, Y. Xiong, B. Lim, S. E. Skrabalak, Angew. Chem. Int. Ed. 2009, 48, 60.
[8] M. Green, Chem. Commun. 2005, 3002
[9] a) A.-V. Mudring, T. Alammar, T. Baecker, K. Richter, ACS Symp. Ser. Ionic Liquids 2009, 1030, 177; b) J. Dupont, G. S. Fonseca, A. P. Umpierre, P. F. P. Fichtner, S. R. Teixeira, J. Am. Chem. Soc. 2002, 124, 4228; c) M. Antonietti, D. Kuang, B. Smarsly, Y. Zhou, Angew. Chem. Int. Ed. 2004, 43, 4988; d) Y. Zhou, Curr. Nanosc. 2005, 1, 35.
Go back: Material Sciences Introduction
For more: Gas phase-based Synthesis