ZMB member Basant Kumar Thakur
ZMB Member
Basant Kumar Thakur
Next Member
Prof. Dr. Basant Kumar Thakur
Group head of
Experimental Molecular and Cellular HepatologyHead Exosome Research Lab,
Department of Gastroenterology, Hepatology and Transplant Medicine
University Hospital Essen
Hufelandstr. 55
45122 Essen
- +49 201 723 2504
- Website
- Press Releases
- Selected Publications
- Publication Metrics
-

- ZMB Research Program
Oncology
Research Overview
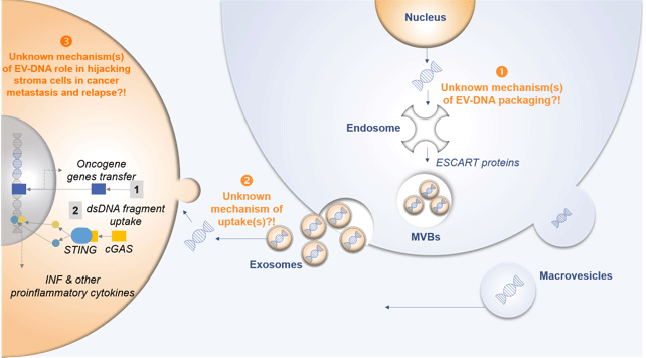
My working group focusses on three key aspects of extracellular vesicles (EV) summarized in the graphical abstract and explained below:
- Identification, purification and characterization of cancer-specific EV carrying the majority of dsDNA with clinical biomarker and functional utility.
- Mechanism(s) of EV dsDNA transfer into the recipient cells.
- Functional consequence of EV dsDNA transfer into recipient cells on metastatic cancer progression.
© B.K. Thakur
Identification and purification of cancer specific EV carrying the majority of dsDNA with clinical biomarker and functional utility
In this project, we aim to dissect the heterogeneity of EVs derived from the mixture of healthy and cancerous cells in the blood plasma of cancer patients in order to obtain EVs with high biomarker potential and functional relevance. We believe that EV surface marker immune capturing-based techniques to enrich DNA-containing EV-subpopulations will further increase the sensitivity of mutational analysis. Using pediatric AML model in collaboration with (Prof. Dr. Bernd Giebel, Transfusion Medicine, UK Essen) we have now established bead based capturing method to isolate and analyze homogenous EV population. Characterization of homogenous leukemia EVs revealed enrichment of leukemia specific mutations in homogenous EV captured by bead capturing. In the next step, we are analyzing a large cohort of pediatric AML and healthy plasma samples for establishing EV DNA based biomarker for minimum residual disease primary responsible for relapse in pediatric patients.
Mechanism of EV dsDNA transfer into the recipient cells
Besides establishing the diagnostic potential of dsDNA in cancer EVs, our lab aims to investigate the mechanism by which tumor EV dsDNA (EV-DNA) is untaken by the healthy recipient cells in the pre-metastatic niche. Key topics related to the study are (i) to understand the molecular pathways explored by the cancer EVs to introduce the DNA inside the recipient cells and (ii) the communication of EV-DNA with the intracellular organelle inside the recipient cells. Detailed functional characterization of the molecular machinery involved in the EV-DNA uptake by distant recipient cells and the interaction of EV-DNA with the cytoplasmic and nuclear components of these recipient cells in the tumor microenvironment will improve our understanding on the biology of cancer metastasis.
Functional consequence of EV dsDNA transfer in recipient cells on metastatic cancer progression
Our data indicating the uptake of EV-DNA by the recipient cells motivates us to explore the function of EV-DNA in pre-metastatic niche immuno-modulation. Our working hypothesis is that EV-DNA modulates immune cell response and can influence the distant metastatic cells towards pro-inflammation. Indeed, our preliminary data suggest the activation of the cytosolic DNA-sensing pathway cGAS/STING (cyclic GMP-AMP synthase—stimulator of interferon genes), resulting in cell cycle arrest or apoptosis in lung fibroblast cells when educated with breast cancer-derived EV-DNA. We believe that increased EV-DNA production by cancer cells and its uptake by immune cells, like dendritic cells in circulation, lead to activation of cGAS/STING signaling. These activated dendritic cells when recruited to the cells in the pre-metastatic niche can induce the whole circuit of inflammatory pathways potentially responsible for the recruitment of tumor cells to the distant organ in the process of metastasis.
Read more
Selected Publications
-
Exploring molecular signatures in PURA syndrome using muscle proteomics and serum biomarkersIn: Journal of Neurology, Vol. 273, 2026, Nr. 2, 94DOI (Open Access)
-
P53 : a new player in the metabolic adaptation of colorectal carcinoma cells under hypoxiaIn: BMC Cancer, Vol. 26, 2026, Nr. 1, 628DOI (Open Access)
-
Single-molecule localization microscopy imaging of extracellular vesicle DNA in recipient cellsIn: Journal of Translational Medicine, Vol. 24, 2026, Nr. 1, 130DOI (Open Access)
-
Blood biomarker fingerprints in a cohort of patients with CHRNE-related congenital myasthenic syndromeIn: Acta Neuropathologica Communications, Vol. 13, 2025, Nr. 1, 29DOI, Online Full Text (Open Access)
-
Cerebrospinal Fluid-Derived Small Extracellular Vesicles May Better Reflect Medulloblastoma Proteomes than Those from Blood PlasmaIn: International Journal of Molecular Sciences (IJMS), Vol. 26, 2025, Nr. 19, 9279DOI (Open Access)
-
Development and In Vitro Characterization of Milk-Derived Extracellular Vesicle-Mithramycin Formulations for Potential Glioma TherapyIn: Molecular Pharmaceutics, Vol. 22, 2025, Nr. 6, pp. 2881 – 2894
-
Fostering the implementation of liquid biopsy in clinical practice : Meeting report 2024 of the European Liquid Biopsy Society (ELBS)In: Journal of Experimental & Clinical Cancer Research, Vol. 44, 2025, Nr. 1, 156DOI (Open Access)
-
Harnessing tomato-derived small extracellular vesicles as drug delivery system for cancer therapyIn: Future Science OA, Vol. 11, 2025, Nr. 1, 2461956DOI (Open Access)
-
Unravelling the Significance of Extracellular Vesicle-Associated DNA in Cancer Biology and Its Potential Clinical ApplicationsIn: Journal of Extracellular Vesicles (JEV), Vol. 14, 2025, Nr. 3, e70047DOI, Online Full Text (Open Access)
-
Intrinsic Burst-Blinking Nanographenes for Super-Resolution BioimagingIn: Journal of the American Chemical Society: JACS, Vol. 146, 2024, Nr. 8, pp. 5195 – 5203DOI (Open Access)
-
Unique structural configuration of EV-DNA primes Kupffer cell-mediated antitumor immunity to prevent metastatic progressionIn: Nature Cancer, Vol. 5, 2024, Nr. 12, pp. 1815 – 1833DOI, Online Full Text (Open Access)
-
Y-box binding protein 1 in small extracellular vesicles reduces mesenchymal stem cell differentiation to osteoblasts : implications for acute myeloid leukaemiaIn: Journal of Extracellular Vesicles (JEV), Vol. 13, 2024, Nr. 3, e12417DOI, Online Full Text (Open Access)
-
Extracellular vesicles transfer chromatin-like structures that induce non-mutational dysfunction of p53 in bone marrow stem cellsIn: Cell Discovery, Vol. 9, 2023, Nr. 1, 12DOI (Open Access)
-
Single Molecule Localization Microscopy for Studying Small Extracellular VesiclesIn: Small, Vol. 19, 2023, Nr. 12, 2205030DOI (Open Access)
-
CD9- and CD81-positive extracellular vesicles provide a marker to monitor glioblastoma cell response to photon-based and proton-based radiotherapyIn: Frontiers in Oncology, Vol. 12, 2022, 947439DOI, Online Full Text (Open Access)
-
DNA in extracellular vesicles : From evolution to its current application in health and diseaseIn: Cell and Bioscience, Vol. 12, 2022, Nr. 1, 37DOI, Online Full Text (Open Access)
-
Efficient Small Extracellular Vesicles (EV) Isolation Method and Evaluation of EV-Associated DNA Role in Cell–Cell Communication in CancerIn: Cancers, Vol. 14, 2022, Nr. 9, 2068DOI (Open Access)
-
Evaluation of Immunoregulatory Biomarkers on Plasma Small Extracellular Vesicles for Disease Progression and Early Therapeutic Response in Head and Neck CancerIn: Cells, Vol. 11, 2022, Nr. 5, 902DOI, Online Full Text (Open Access)
-
Glioma : molecular signature and crossroads with tumor microenvironmentIn: Cancer and Metastasis Reviews, Vol. 41, 2022, pp. 53 – 75DOI (Open Access)
-
Single extracellular vesicle analysis performed by imaging flow cytometry and nanoparticle tracking analysis evaluate the accuracy of urinary extracellular vesicle preparation techniques differentlyIn: International Journal of Molecular Sciences (IJMS), Vol. 22, 2021, Nr. 22, 12436DOI, Online Full Text (Open Access)
-
Evaluation of dsDNA from extracellular vesicles (EVs) in pediatric AML diagnosticsIn: Annals of Hematology, Vol. 99, 2020, Nr. 3, pp. 459 – 475
-
Insights into the limitations of transient expression systems for the functional study of p53 acetylation site and oncogenic mutantsIn: Biochemical and Biophysical Research Communications (BBRC), Vol. 524, 2020, Nr. 4, pp. 990 – 995
-
Tumor-Derived Extracellular Vesicles and the Immune System : Lessons From Immune-Competent Mouse-Tumor ModelsIn: Frontiers in Immunology, Vol. 11, 2020, pp. 606859DOI (Open Access)
-
Tumour progression stage-dependent secretion of yb-1 stimulates melanoma cell migration and invasionIn: Cancers, Vol. 12, 2020, Nr. 8, pp. 1 – 17DOI (Open Access)
-
Challenges in the Isolation and Proteomic Analysis of Cancer Exosomes : Implications for Translational ResearchIn: Proteomes, Vol. 7, 2019, Nr. 2, 22DOI, Online Full Text (Open Access)
-
Detection of AML-specific mutations in pediatric patient plasma using extracellular vesicle–derived RNAIn: Annals of Hematology, Vol. 98, 2019, Nr. 3, pp. 595 – 603
-
Effects of Irradiation with Protons and Photons on the Survival of Glioblastoma Cell Lines and the Secretion of Exosomes in vitroIn: Strahlentherapie und Onkologie: Journal of Radiation Oncology, Biology, Physics, Vol. 195, 2019, Nr. Suppl. 1, pp. 173
-
NAMPT signaling is critical for the proangiogenic activity of tumor-associated neutrophilsIn: International Journal of Cancer, Vol. 144, 2019, Nr. 1, pp. 136 – 149
-
Packaging and transfer of mitochondrial DNA via exosomes regulate escape from dormancy in hormonal therapy-resistant breast cancerIn: Proceedings of the National Academy of Sciences of the United States of America (PNAS), Vol. 114, 2017, Nr. 43, pp. E9066 – E9075
-
Extracellular Vesicles in Cancer : Cell-to-Cell Mediators of MetastasisIn: Cancer Cell, Vol. 30, 2016, Nr. 6, pp. 836 – 848DOI, Online Full Text (Open Access)

