News

02.11.2022Gottschalk-Diederich-Baedeker-Award for Corina Andronescu
Our TRR 247 member Corina Andronescu was awarded the Gottschalk-Diederich-Baedeker-Award - Congratulations!
After the award ceremony, Corina gave her inaugural lecture at the UDE entitled: “Electrochemistry in Energy Conversion and Storage. From macro to nano scale“.
Read the UDE press release here.

19.10.20228th Ruhr-Symposium: Functional Materials for Hydrogen
Functional materials for hydrogen are the focus of the 8th Ruhr-Symposium. In tandem talks, experts from industrial and academic partnerships will discuss concepts, synergies and scientific results.
A special feature of this year’s event is the scheduling of the Ruhr-Symposium together with the Duisburg Future Talks, a public panel discussion sponsored by the MERCATOR Foundation and organized by Duisburg Business & Innovation and the Hy.Region.Rhein.Ruhr association, which will take place as an evening event.
Between the symposium and the Duisburg Future Talks there will be an interesting bridge program, where we invite representatives from industry and the public to visit the NETZ building and the ZBT.
The event will take place at the Fraunhofer inHaus Center at the campus Duisburg.
More information and registration are available here.

05./06.10.2022PhD Retreat
The first PhD Retreat of the new funding period took place 5th-6th October 2022.
All new and experienced PhD students of TRR 247 participated in this 1.5-days event. To break the ice, the event started with a joint visit of an Escape Room in Bochum. The second day was all about networking. The PhD students presented their projects in elevator pitches and first collaborations were established. In the afternoon, the students visited the labs of our TRR 247 members Prof. Tschulik and Prof. Muhler.

04./05.10.2022Kick-off Meeting in Bochum
The Kick-off meeting of the second funding period took place at the Ruhr University Bochum on 4th-5th October 2022.
The entire consortium, including project leaders and young researchers, participated in this two-days event. Besides impulse-sessions of our three Research Areas A - Catalysis, B - Analysis, and C - Synthesis, our new members presented their projects and fruitful discussions took place about different research topics and projects of our TRR 247.

15.08.2022African fictions to boost the scientific culture in Africa
Dr. Stéphane Kenmoe, member of our CRC/TRR 247, is working as a postdoctoral researcher at the Chair of Theoretical Chemistry at the University of Duisburg-Essen. Born in Cameroon, he is actively involved in promoting African early career researchers and communicate popular scientific topics in an easily understandable way to the African community. In his home country, he is known for TV appearances in which he explains scientific phenomena in a generally understandable and humorous way.

01.07.2022Soft Skill Summer 2022
The Soft Skills Summer is an initiative by the CRC/TRR 247, the CRC 1242, CENIDE, the SPP 1980, FOR 2284 and the team for Equal Opportunities of the Faculty of Physics at the University of Duisburg-Essen. We would like to invite all female PhD candidates and postdocs to be a part of it and to improve your soft skills.

30.05.2022Second funding period granted for TRR 247
Congratulations!!
TRR 247 receives funding from the German Research Foundation (DFG) for a second funding period of another four years (2022-2026).

March 2022Research Opportunities for Ukrainian Researchers
In order to express our solidarity with the people in Ukraine, CRC/TRR 247 provides funding for research stays and visiting professors of up to 6 months for Ukrainian scientists to be hosted by one of our research groups.
Are you interested in completing a research project in the interdisciplinary field of oxidation catalysis? Check out the CRC/TRR 247 website to get more information on the involved groups and disciplines. Please get in contact with us via info@trr247.de and send us your CV and one-page outline of possible projects within CRC/TRR 247.
Further information here.

15.03.2022Prof. Kai Exner awarded with Aduc Prize
Jun.-Prof. Dr. rer. nat. Kai Steffen Exner, University Duisburg-Essen, Germany is awarded with the ADUC prize 2022 (Arbeitsgemeinschaft Deutscher Universitätsprofessoren und -professorinnen für Chemie, ADUC).
As part of the Chemiedozententagung , the ADUC awards the ADUC prize to up to three young scientists (during their habilitation, scholarship or junior professorship) for establishing an independent research area. The award is combined with an award certificate and prize money of 5,000 Euros each. The establishment of the independent research area is to be demonstrated by means of publications in scientific journals.
Congratulations!
The respective press release you can find here.

08.03.2022Prof. Kai Exner awarded with Jochen Block Prize
The Jochen Block Prize 2022 of the German Catalysis Society (GeCatS) goes to Jun.-Prof. Dr. rer. nat. Kai Steffen Exner, University Duisburg-Essen / Germany. It recognizes his original and pioneering contributions in the field of theoretical electrocatalysis. The Jochen Block Prize honours the achievements of young scientists. It is endowed with 3,000 Euros and will be presented at the Annual German Catalysis Meeting on March 17, 2022 in Weimar / Germany.
Exner and his research group are working on electrochemical reactions on solid-state catalysts. These processes play a key role for a sustainable energy supply, for instance to split water into gaseous oxygen and the energy carrier gaseous hydrogen. Exner has developed new methods to predict the performance of electrodes, which helps to identify promising combinations of catalyst materials as well as to optimize existing materials. In this way, complex and time-consuming “trial and error” tests can be avoided. In his latest research work, Exner has used concepts from catalysis to optimize drug-delivery systems for cancer therapy. He has thus built a bridge between catalysis and medical research, which also provides new impulses for the scientific discipline of theoretical electrochemistry.
Congratulations!
You can find the respective press release here

10.01.2022Nature Communications: Catalyst Surface Analyzed at Atomic Resolution
A German-Chinese research team has visualized the three-dimensional structure of the surface of catalyst nanoparticles at atomic resolution. This structure plays a decisive role in the activity and stability of the particles.
Researchers from the Ruhr-Universität Bochum, the University of Duisburg-Essen and the Max Planck Institute for Chemical Energy Conversion in Mülheim an der Ruhr cooperated on the project as part of the Collaborative Research Centre “Heterogeneous oxidation catalysis in the liquid phase".
At RUB, a team headed by Weikai Xiang and Professor Tong Li from Atomic-scale Characterisation worked together with the Chair of Electrochemistry and Nanoscale Materials and the Chair of Industrial Chemistry. Institutes in Shanghai, China, and Didcot, UK, were also involved. The team presents their findings in the journal Nature Communications.
You can find the respective press release here

03.11.20217th RUHR-Symposium: Functional Materials for Catalysis
Functional materials for catalysis are focused on the 7th RUHR Symposium. In tandem talks, experts from industrial and academic partnerships will discuss concepts, synergies and scientific results.
More information is available here

01.10.2021Berthold Leibinger Innovation Prize 2021
Prof. Stephan Barcikowski (Univ. Duisburg-Essen) and his colleague Prof. Bilal Gökce (Univ. Wuppertal) had been awarded the 3rd prize of the Berthold Leibinger Innovation Prize 2021. The award is one of the most important and most highly endowed innovation and research prizes in optics and laser research and is presented every two years to scientists who break new ground in the application of laser technology. The team was awarded for its research on Scaled-up Nanoparticle Synthesis by Laser Ablation in Liquids for the Chemical Industry and Additive Manufacturing.
Congratulations!
The respective UDE article is available here.

27./28.09.2021Annual CRC/TRR 247 meeting
Venue: online
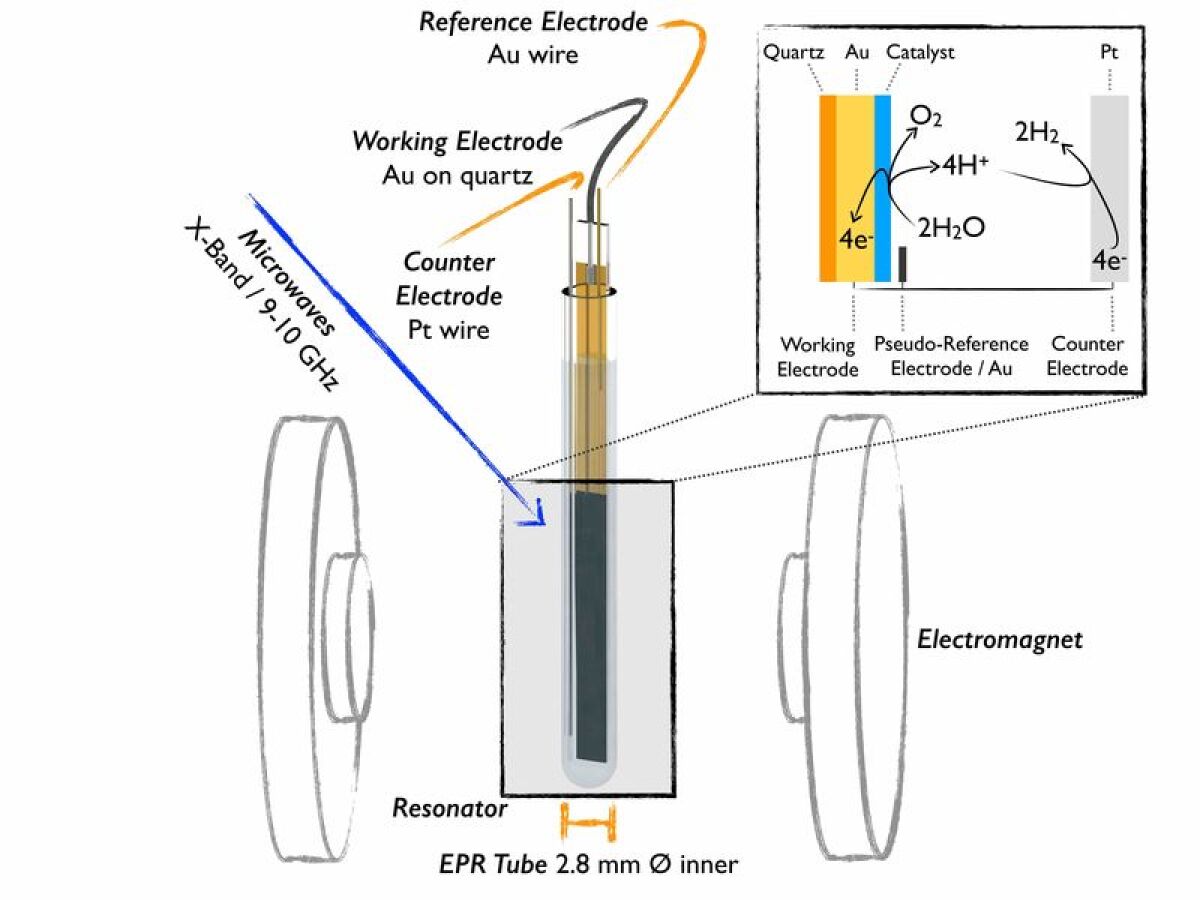
14 September 2021UnOCat Electron Paramagnetic Resonance Spectroscopy Workshop
Our workshop on "Electron Paramagnetic Resonance Spectroscopy" will take place online between 9:30 am and 12:15 pm.
The agenda is available here.
If you like to participate, please send an E-Mail to the coordinator.

Summer 2021MGK Summer
The MGK Summer is an initiative by the CRC 1242, CRC/TRR 270 Hommage und CRC/TRR 247.
We would like to invite our PhD students to be a part of our first online MGK Summer to improve their soft skills.
Further information is available here.

Summer 2021Soft Skills Summer: Women in Academia
The Soft Skills Summer is an initiative by the CRC 1242, CRC/TRR 270 Hommage und CRC/TRR 247 and the team for Equal Opportunities of the Faculty of Physics at the University of Duisburg-Essen. We would like to invite you to be a part of it and to improve your soft skills.
Further information is available here.

27.08.2021Stéphane Kenmoe - Falling Walls Engage Winner 2021
Falling Walls Engage is the global platform for Science Engagement, hosted by the Falling Walls Foundation in cooperation with the Robert Bosch Stiftung. Their mission is to inspire and connect creative individuals and organisations who actively involve the public with science to generate mutual benefits for science and society all around the world.
Stéphane is one of 20 Winners (189 applications) who was selected by an Advisory Board. He will pitch his science engagement project on stage on November 7, 2021 in front of an international Jury in Berlin to become the Breakthrough of the Year in the category Science Engagement.
Congratulations, Stéphane, and good luck!
For more details about his engagement, watch the interview.

16.08.2021Twitter: @ExnerKai
Follow Kai Exner lab on Twitter:

29.07.2021Synergic publication in high ranked journal
We are happy to announce our most recent cross-project publication, including PIs from all of our three research areas: A6, B6, C1, and C5.
The publication reports nanosecond, single-pulse laser post-processing (PLPP) in a liquid flat jet with precise control of the applied laser intensity to tune structure, defect sites, and the oxygen evolution reaction (OER) activity of mesostructured Co3O4.
The work was published in ACS Applied Materials and Interfaces.
Congratulations!
The publication is available here.

19.07.2021Prof. Kai Exner awarded with Max-Buchner Research Scholarship 2021
Prof. Kai Exner was awarded with the Max-Buchner Research Scholarship for his project "Identifying the optimum hydrogen binding energy of hydrogenases". The Max Buchner Research Foundation was established in 1936 and was named after the founder of DECHEMA. The foundation supports research through the award of scholarships in the fields of chemical engineering, process engineering, biotechnology and environmental protection.
Congratulations!
A press release (in German) is available here.

13.07.2021Prof. Kai Exner awarded with Ewald-Wicke Prize 2021
Prof. Kai Exner was awarded the renowned Ewald-Wicke Prize. The German Bunsen Society for Physical Chemistry (DBG) and the Ewald-Wicke-Foundation honored his pioneering research on the description of elementary processes in batteries and electrolyzers by modern theoretical methods and the concept of free-energy diagram. Prof. Exner accepted the award at the DBG's digital general assembly in mid-May.
Congratulations!
Further information is available here.

12.-16.07.2021UnOCat Summer School
The first Summer School, which was supposed to take place in July 2020, has to be postponed to July 2021 because of Covid-19. The Summer School will take place online.
Further information is available here.

Summer Semester 2021Joint CataLysis Online Colloquium Series
Our Joint CataLysis Online Colloquium Series will start with the lecture of Prof. Yang Shao-Horn, MIT, on 29th of June.
Further information is available here. More information about the CataLysis network is available here.
If you're interested in participating, please send a mail to F. Günther to get the login information.

15.06.2021UnOCat Spray Flame Synthesis Workshop
Our workshop on "Spray Flame Synthesis" will take place online between 9:30 am and 2 pm.
More information is available here.
The workshop is organised by CRC/TRR 247 and the DFG Priority Program SPP1980 (Nanoparticle Synthesis in Spray Flames).

01.06.2021NRW returnee receives junior professorship
His work benefits battery research as well as medicine: Dr. Kai S. Exner will become junior professor for Theoretical Inorganic Chemistry at the UDE starting in June.
Exner studies electrochemical reactions on solid-state catalysts. He has developed his own theory that can be used to predict the performance of such catalyst materials. But it can also be used to make statements about how to optimize existing materials.
The research group that Exner is currently setting up will focus in particular on inorganic materials and their use for electrochemical reactions. In addition, he will strengthen cutting-edge research at the UDE as well as within the University Alliance Ruhr: Exner has been accepted into the Center for Nanointegration (CENIDE). He also collaborates in the Collaborative Research Center 247 "Heterogeneous Oxidation Catalysis in the Liquid Phase" and in the Cluster of Excellence RESOLV (Ruhr Explores Solvation).
> more about: Press release University of Duisburg-Essen, 01.06.2021

29.04.2021UnOCat "Electron Microscopy Workshop"
Our UnOCat Electron Microscopy Workshop will take place online.
The lecturers are:
- Dr. Thomas Götsch, FHI
- Dr. Franz Schmidt, FHI
- Dr. Thomas Lunkenbein, FHI

22.04.2021MYSTIIC: New X-ray microscope at BESSY II
A new X-ray microscope has started operation at the Energy Materials in situ Lab (EMIL) in collaboration with the Inorganic Chemistry Department of the Fritz-Haber-Institut. It is a scanning transmission X-ray microscope designed to examine both sample surfaces and bulk sample. With the soft X-ray light from BESSY II, it is even possible to localise individual elements and chemical compounds.
Press release Fritz Haber Institute, 22.04.2021

MondaysOnline Lunch Meetings
We use our weekly lunch meetings on Monday from 12 to 1 pm to discuss specific research topics with all interested members of the CRC/TRR 247. On 26th of April, we start on the topic "Materials Chemistry".
Members of the CRC/TRR 247 can find further information in our CRC database.

20.04.2021CRC/TRR 247 Retreat II
The follow-up meeting of the PIs and Postdocs will also take place online.

Publication AlertStable and Catalytically Active Pigment
It is one of the softest white pigments used by the industry. However, zinc sulfide turns gray over time if it is not appropriately pretreated. Members of the CRC/TRR 247 discovered a way to retain its brilliant color and also enable its use as a catalyst; for example, to convert sunlight into usable energy. The scientific journal "Advanced Functional Materials" covers the topic in its latest issue.
Original publication:
Lange, T., Reichenberger, S., Rohe, M., Bartsch, M., Kampermann, L., Klein, J., Strunk, J., Bacher, G., Schlögl, R., Barcikowski, S: Alumina-Protected, Durable and Photostable Zinc Sulfide Particles from Scalable Atomic Layer Deposition. Adv. Funct. Mater. 2021, 2009323. DOI: 10.1002/adfm.202009323.
> more about: Press release CENIDE, University of Duisburg-Essen, 24.03.2021

22./23.03.20212. PhD Retreat
The second UnOCat retreat will take place online due to the COVID-19 pandemic.
On the second day of the retreat, there will be a workshop on "Good Scientific Practice".

Publication alertPredicting the Performance of Catalysts
Green electricity should be available as soon as possible, at best from all power outlets: electrochemical processes are the basis for a sustainable energy, but they need new, high-performance catalyst materials. Theoretical calculations help to avoid dead ends and to focus on the most promising candidates. Dr. Kai Exner, associated member of the CRC/TRR 247, has decisively improved this performance prediction and has now received an award for his achievements. He published his work in the trade journal Electrochimica Acta.
Original publication:
Exner, K.S.: Why approximating electrocatalytic activity by a single free-energy change is insufficient. Electrochim. Acta 2021, 375, 137975. DOI: 10.1016/j.electacta.2021.137975.
> more about: Press release CENIDE, University of Duisburg-Essen, 11.03.2021

24.02.2021UnOCat Lecture Series "Photocatalysis"
Our UnOCat Lecture Series on "Electrocatalysis" will take place online.
The lecturers are:
- Prof. Martin Muhler, Ruhr-University Bochum
- Prof. Lilac Amirav, Technion - Israel Institute of Technology
- Prof. Gerd Bacher, University of Duisburg-Essen
- Prof. Guido Mul, University of Twente
If you're interested in participating, please send a mail to F. Günther to get the login information.

Publication alertDiscovery of New Solid Catalysts for Water Electrolysis
PD Dr. Harun Tüysüz (Max-Planck-Institut für Kohlenforschung) and colleagues have discovered a new type of OER electrocatalyst. A variety of Co2YZ type Heusler compounds with tunable physicochemical properties and well-defined topological surfaces were designed and demonstrated to effectively split water into oxygen and hydrogen.
This work demonstrates proof of concept implementation of Heusler compounds as a new class of OER electrocatalysts, and the effect of orbital occupation on their catalytic performances. The results of this study were published in Angewandte Chemie International Edition as “Hot Paper”.
Original publication:
Yu, M., Li, G., Fu, C., Liu, E., Manna, K., Budiyanto, E., Yang, Q., Felser, C., Tüysüz, H.: Tunable eg Orbital Occupancy in Heusler Compounds for Oxygen Evolution Reaction, Angew. Chem. Int. Ed. 2021, 60, 1 – 7. DOI: 10.1002/anie.202013610.
> more about: Press release Max-Planck-Institut für Kohlenforschung, 22.02.2021
> more about: Press release Chemistry Views, 22.02.2021

10./11.02.2021CRC/TRR 247 Retreat I
Due to the COVID-19 pandemic, our retreat will take place online.
Participants are all PIs and Postdocs.


